|
Synonyms: ONC-201 | TIC-10 | TIC10
Compound class:
Synthetic organic
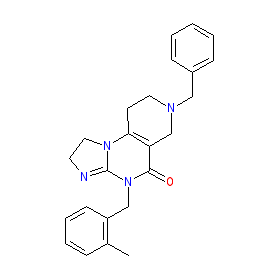
Comment: ONC201 (also known as TIC10 [2]) was originally discovered as a TRAIL receptor inducer and as an orally active dual Akt/ERK inhibitor [1] It exerts potent anti-tumour effects.
Note that although [1] and [2] are published by the same research group, both articles show different isomers of the chemical structure. We show the structure from their 2016 paper, which introduces ONC201 as the first-in-class imipridone anti-cancer compound [1]. PubChem CID 336423 represents the alternative isomer as depicted in the 2013 paper [2]. ONC201 was later reported as an allosteric activator of the mitochondrial protease caseinolytic protease P (ClpP) [4]. ClpP is an oncology drug target [3]. |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Allen JE, Kline CL, Prabhu VV, Wagner J, Ishizawa J, Madhukar N, Lev A, Baumeister M, Zhou L, Lulla A et al.. (2016)
Discovery and clinical introduction of first-in-class imipridone ONC201. Oncotarget, 7 (45): 74380-74392. [PMID:27602582] |
|
2. Allen JE, Krigsfeld G, Mayes PA, Patel L, Dicker DT, Patel AS, Dolloff NG, Messaris E, Scata KA, Wang W et al.. (2013)
Dual inactivation of Akt and ERK by TIC10 signals Foxo3a nuclear translocation, TRAIL gene induction, and potent antitumor effects. Sci Transl Med, 5 (171): 171ra17. [PMID:23390247] |
|
3. Ishizawa J, Zarabi SF, Davis RE, Halgas O, Nii T, Jitkova Y, Zhao R, St-Germain J, Heese LE, Egan G et al.. (2019)
Mitochondrial ClpP-Mediated Proteolysis Induces Selective Cancer Cell Lethality. Cancer Cell, 35 (5): 721-737.e9. [PMID:31056398] |
|
4. Prabhu VV, Morrow S, Rahman Kawakibi A, Zhou L, Ralff M, Ray J, Jhaveri A, Ferrarini I, Lee Y, Parker C et al.. (2020)
ONC201 and imipridones: Anti-cancer compounds with clinical efficacy. Neoplasia, 22 (12): 725-744. [PMID:33142238] |
|
5. Wagner J, Kline CL, Zhou L, Campbell KS, MacFarlane AW, Olszanski AJ, Cai KQ, Hensley HH, Ross EA, Ralff MD et al.. (2018)
Dose intensification of TRAIL-inducing ONC201 inhibits metastasis and promotes intratumoral NK cell recruitment. J Clin Invest, 128 (6): 2325-2338. [PMID:29533922] |