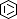kinase insert domain receptor
Target not currently curated in GtoImmuPdb
Target id: 1813
Nomenclature: kinase insert domain receptor
Abbreviated Name: VEGFR-2
Family: Type IV RTKs: VEGF (vascular endothelial growth factor) receptor family
Contents:
- Gene and Protein Information
- Previous and Unofficial Names
- Database Links
- Selected 3D Structures
- Enzyme Reaction
- Natural/Endogenous Ligands
- Inhibitors
- Agonists
- Antibodies
- Large-scale Ligand Screening Data
- Immuno Process Associations
- Clinically-Relevant Mutations and Pathophysiology
- General Comments
- References
- How to cite this page
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | 1 | 1356 | 4q12 | KDR | kinase insert domain receptor | |
| Mouse | 1 | 1367 | 5 40.23 cM | Kdr | kinase insert domain protein receptor | |
| Rat | 1 | 1343 | 14p11 | Kdr | kinase insert domain receptor | |
Previous and Unofficial Names  |
| CD309 | fetal liver kinase 1 | FLK1 | FLK4 | kinase insert domain receptor (a type III receptor tyrosine kinase) | vascular endothelial growth factor receptor 2 |
Database Links  |
|
| Alphafold | P35968 (Hs), P35918 (Mm), O08775 (Rn) |
| BRENDA | 2.7.10.1 |
| CATH/Gene3D | 2.60.40.10 |
| ChEMBL Target | CHEMBL279 (Hs), CHEMBL3337 (Mm) |
| DrugBank Target | P35968 (Hs) |
| Ensembl Gene | ENSG00000128052 (Hs), ENSMUSG00000062960 (Mm), ENSRNOG00000046829 (Rn) |
| Entrez Gene | 3791 (Hs), 16542 (Mm), 25589 (Rn) |
| Human Protein Atlas | ENSG00000128052 (Hs) |
| KEGG Enzyme | 2.7.10.1 |
| KEGG Gene | hsa:3791 (Hs), mmu:16542 (Mm), rno:25589 (Rn) |
| OMIM | 191306 (Hs) |
| Orphanet | ORPHA229801 (Hs) |
| Pharos | P35968 (Hs) |
| RefSeq Nucleotide | NM_002253 (Hs), NM_010612 (Mm), NM_013062 (Rn) |
| RefSeq Protein | NP_002244 (Hs), NP_034742 (Mm), NP_037194 (Rn) |
| SynPHARM |
78759 (in complex with axitinib) 81169 (in complex with axitinib) 79689 (in complex with lenvatinib) 79677 (in complex with nintedanib) 81175 (in complex with sorafenib) 81305 (in complex with sorafenib) 81287 (in complex with sunitinib) 79680 (in complex with tivozanib) |
| UniProtKB | P35968 (Hs), P35918 (Mm), O08775 (Rn) |
| Wikipedia | KDR (Hs) |
Selected 3D Structures  |
|||||||||||||
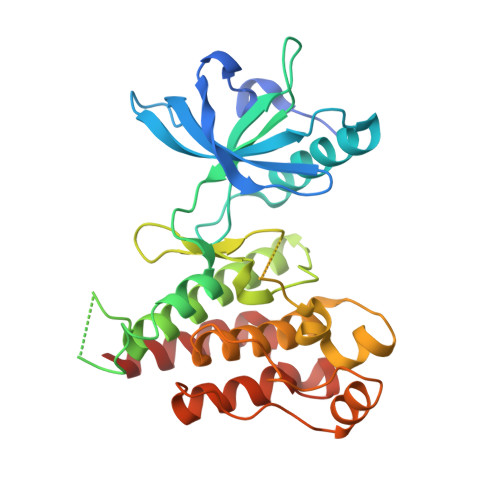
|
|
||||||||||||
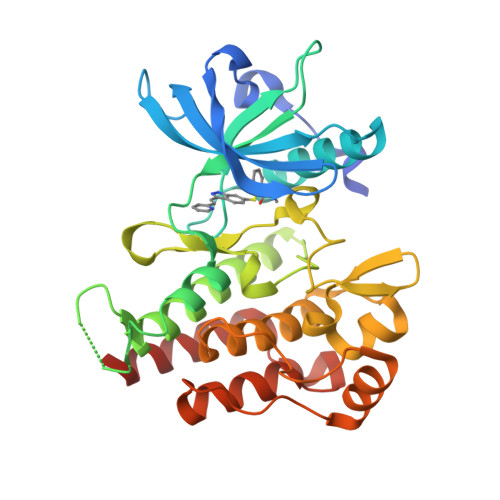
|
|
||||||||||||
Enzyme Reaction  |
||||
|
||||
Natural/Endogenous Ligands  |
| VEGFA {Sp: Human} |
| VEGFC {Sp: Human} |
| VEGFE {Sp: Human} |
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| View species-specific inhibitor tables | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inhibitor Comments | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note that pazopanib is a multi-target kinase inhibitor. See [31] for additional data. Vandetanib also inhibits EGFR and RET tyrosine kinases, as well as VEGFR2. Although LY2874455 has equipotent IC50s for FGFRs and VEGFR2 in biochemical assays, in vivo efficacy appears to be 6-9-fold less for VEGFR2 phosphorylation in mouse heart muscle compared to inhibition of FGFR phosphorylation [87]. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Agonists | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Agonist Comments | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Note that a VEGFA mimetic probe peptide (scVEGF) was used to determine the dissociation constants in the table above. | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Antibodies | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DiscoveRx KINOMEscan® screen  |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A screen of 72 inhibitors against 456 human kinases. Quantitative data were derived using DiscoveRx KINOMEscan® platform. http://www.discoverx.com/services/drug-discovery-development-services/kinase-profiling/kinomescan Reference: 16,82 |
 
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Target used in screen: VEGFR2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displaying the top 10 most potent ligands View all ligands in screen » | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
EMD Millipore KinaseProfilerTM screen/Reaction Biology Kinase HotspotSM screen  |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A screen profiling 158 kinase inhibitors (Calbiochem Protein Kinase Inhibitor Library I and II, catalogue numbers 539744 and 539745) for their inhibitory activity at 1µM and 10µM against 234 human recombinant kinases using the EMD Millipore KinaseProfilerTM service. A screen profiling the inhibitory activity of 178 commercially available kinase inhibitors at 0.5µM against a panel of 300 recombinant protein kinases using the Reaction Biology Corporation Kinase HotspotSM platform. http://www.millipore.com/techpublications/tech1/pf3036 http://www.reactionbiology.com/webapps/main/pages/kinase.aspx Reference: 4,25 |

|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Target used in screen: KDR/KDR(VEGFR2) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displaying the top 10 most potent ligands View all ligands in screen » | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immuno Process Associations | ||
|
||
|
Clinically-Relevant Mutations and Pathophysiology 
|
||||||||||||
|
||||||||||||
| General Comments |
| This protein contains an immunoglobulin (Ig)-like domain that resembles the antibody variable domain, that has been coined the 'V-set domain'. The genes for all human V-set domain containing proteins are listed in HGNC gene group 590. |
References
1. Adams CM, Anderson K, Artman 3rd G, Bizec JC, Cepeda R, Elliott J, Fassbender E, Ghosh M, Hanks S, Hardegger LA et al.. (2018) The Discovery of N-(1-Methyl-5-(trifluoromethyl)-1H-pyrazol-3-yl)-5-((6- ((methylamino)methyl)pyrimidin-4-yl)oxy)-1H-indole-1-carboxamide (Acrizanib), a VEGFR-2 Inhibitor Specifically Designed for Topical Ocular Delivery, as a Therapy for Neovascular Age-Related Macular Degeneration. J Med Chem, 61 (4): 1622-1635. [PMID:29400470]
2. Akeno-Stuart N, Croyle M, Knauf JA, Malaguarnera R, Vitagliano D, Santoro M, Stephan C, Grosios K, Wartmann M, Cozens R et al.. (2007) The RET kinase inhibitor NVP-AST487 blocks growth and calcitonin gene expression through distinct mechanisms in medullary thyroid cancer cells. Cancer Res, 67 (14): 6956-64. [PMID:17638907]
3. Albert DH, Tapang P, Magoc TJ, Pease LJ, Reuter DR, Wei RQ, Li J, Guo J, Bousquet PF, Ghoreishi-Haack NS et al.. (2006) Preclinical activity of ABT-869, a multitargeted receptor tyrosine kinase inhibitor. Mol Cancer Ther, 5 (4): 995-1006. [PMID:16648571]
4. Anastassiadis T, Deacon SW, Devarajan K, Ma H, Peterson JR. (2011) Comprehensive assay of kinase catalytic activity reveals features of kinase inhibitor selectivity. Nat Biotechnol, 29 (11): 1039-45. [PMID:22037377]
5. Apsel B, Blair JA, Gonzalez B, Nazif TM, Feldman ME, Aizenstein B, Hoffman R, Williams RL, Shokat KM, Knight ZA. (2008) Targeted polypharmacology: discovery of dual inhibitors of tyrosine and phosphoinositide kinases. Nat Chem Biol, 4 (11): 691-9. [PMID:18849971]
6. AstraZeneca. AZD4547. Accessed on 11/09/2014. Modified on 11/09/2014. astrazeneca.com, http://openinnovation.astrazeneca.com/what-we-offer/compound/azd4547/
7. Barker MD, Liddle J, Atkinson FL, Wilson DM, Dickson MC, Ramirez-Molina C, Lewis H, Davis RP, Somers DO, Neu M et al.. (2018) Discovery of potent and selective Spleen Tyrosine Kinase inhibitors for the topical treatment of inflammatory skin disease. Bioorg Med Chem Lett, 28 (21): 3458-3462. [PMID:30249354]
8. Beebe JS, Jani JP, Knauth E, Goodwin P, Higdon C, Rossi AM, Emerson E, Finkelstein M, Floyd E, Harriman S et al.. (2003) Pharmacological characterization of CP-547,632, a novel vascular endothelial growth factor receptor-2 tyrosine kinase inhibitor for cancer therapy. Cancer Res, 63 (21): 7301-9. [PMID:14612527]
9. Bhide RS, Cai ZW, Zhang YZ, Qian L, Wei D, Barbosa S, Lombardo LJ, Borzilleri RM, Zheng X, Wu LI et al.. (2006) Discovery and preclinical studies of (R)-1-(4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-5- methylpyrrolo[2,1-f][1,2,4]triazin-6-yloxy)propan- 2-ol (BMS-540215), an in vivo active potent VEGFR-2 inhibitor. J Med Chem, 49 (7): 2143-6. [PMID:16570908]
10. Bold G, Altmann KH, Frei J, Lang M, Manley PW, Traxler P, Wietfeld B, Brüggen J, Buchdunger E, Cozens R et al.. (2000) New anilinophthalazines as potent and orally well absorbed inhibitors of the VEGF receptor tyrosine kinases useful as antagonists of tumor-driven angiogenesis. J Med Chem, 43 (12): 2310-23. [PMID:10882357]
11. Chen GP. (2008) Spiro substituted compounds as angiogenesis inhibitors. Patent number: WO2008112407 A1. Assignee: Advenchen Laboratories, Llc. Priority date: 14/03/2007. Publication date: 18/09/2008.
12. Cho TP, Dong SY, Jun F, Hong FJ, Liang YJ, Lu X, Hua PJ, Li LY, Lei Z, Bing H et al.. (2010) Novel potent orally active multitargeted receptor tyrosine kinase inhibitors: synthesis, structure-activity relationships, and antitumor activities of 2-indolinone derivatives. J Med Chem, 53 (22): 8140-9. [PMID:21028894]
13. Claridge S, Raeppel F, Granger MC, Bernstein N, Saavedra O, Zhan L, Llewellyn D, Wahhab A, Deziel R, Rahil J et al.. (2008) Discovery of a novel and potent series of thieno[3,2-b]pyridine-based inhibitors of c-Met and VEGFR2 tyrosine kinases. Bioorg Med Chem Lett, 18 (9): 2793-8. [PMID:18434145]
14. Conesa-Milián L, Falomir E, Murga J, Carda M, Marco JA. (2019) Novel multitarget inhibitors with antiangiogenic and immunomodulator properties. Eur J Med Chem, 170: 87-98. [PMID:30878834]
15. Curtin ML, Heyman HR, Frey RR, Marcotte PA, Glaser KB, Jankowski JR, Magoc TJ, Albert DH, Olson AM, Reuter DR et al.. (2012) Pyrazole diaminopyrimidines as dual inhibitors of KDR and Aurora B kinases. Bioorg Med Chem Lett, 22 (14): 4750-5. [PMID:22695126]
16. Davis MI, Hunt JP, Herrgard S, Ciceri P, Wodicka LM, Pallares G, Hocker M, Treiber DK, Zarrinkar PP. (2011) Comprehensive analysis of kinase inhibitor selectivity. Nat Biotechnol, 29 (11): 1046-51. [PMID:22037378]
17. Dominguez C, Smith L, Huang Q, Yuan C, Ouyang X, Cai L, Chen P, Kim J, Harvey T, Syed R et al.. (2007) Discovery of N-phenyl nicotinamides as potent inhibitors of Kdr. Bioorg Med Chem Lett, 17 (21): 6003-8. [PMID:17869515]
18. Dumas JP, Joe TK, Kluender HCE, Lee W, Nagarantham D, Sibley RN, Su S, Boyer SJ, Dixon JA. (2001) Substituted pyridines and pyridazines with angiogenesis inhibiting activity. Patent number: WO2001023375A2. Assignee: Bayer Corporation. Priority date: 28/09/1999. Publication date: 05/04/2001.
19. Engers DW, Frist AY, Lindsley CW, Hong CC, Hopkins CR. (2013) Synthesis and structure-activity relationships of a novel and selective bone morphogenetic protein receptor (BMP) inhibitor derived from the pyrazolo[1.5-a]pyrimidine scaffold of dorsomorphin: the discovery of ML347 as an ALK2 versus ALK3 selective MLPCN probe. Bioorg Med Chem Lett, 23 (11): 3248-52. [PMID:23639540]
20. Fedorov O, Marsden B, Pogacic V, Rellos P, Müller S, Bullock AN, Schwaller J, Sundström M, Knapp S. (2007) A systematic interaction map of validated kinase inhibitors with Ser/Thr kinases. Proc Natl Acad Sci USA, 104 (51): 20523-8. [PMID:18077363]
21. Feldman RI, Wu JM, Polokoff MA, Kochanny MJ, Dinter H, Zhu D, Biroc SL, Alicke B, Bryant J, Yuan S et al.. (2005) Novel small molecule inhibitors of 3-phosphoinositide-dependent kinase-1. J Biol Chem, 280 (20): 19867-74. [PMID:15772071]
22. Flynn DL, Kaufman MD, Smith BD. (2016) Inhibition of tumor cell interactions with the microenvironment resulting in a reduction in tumor growth and disease progression. Patent number: WO2016061231. Assignee: Deciphera Pharmaceuticals, Llc. Priority date: 14/10/2014. Publication date: 21/04/2016.
23. Fraley ME, Rubino RS, Hoffman WF, Hambaugh SR, Arrington KL, Hungate RW, Bilodeau MT, Tebben AJ, Rutledge RZ, Kendall RL et al.. (2002) Optimization of a pyrazolo[1,5-a]pyrimidine class of KDR kinase inhibitors: improvements in physical properties enhance cellular activity and pharmacokinetics. Bioorg Med Chem Lett, 12 (24): 3537-41. [PMID:12443771]
24. Fujita H, Miyadera K, Kato M, Fujioka Y, Ochiiwa H, Huang J, Ito K, Aoyagi Y, Takenaka T, Suzuki T et al.. (2013) The novel VEGF receptor/MET-targeted kinase inhibitor TAS-115 has marked in vivo antitumor properties and a favorable tolerability profile. Mol Cancer Ther, 12 (12): 2685-96. [PMID:24140932]
25. Gao Y, Davies SP, Augustin M, Woodward A, Patel UA, Kovelman R, Harvey KJ. (2013) A broad activity screen in support of a chemogenomic map for kinase signalling research and drug discovery. Biochem J, 451 (2): 313-28. [PMID:23398362]
26. Garton AJ, Crew AP, Franklin M, Cooke AR, Wynne GM, Castaldo L, Kahler J, Winski SL, Franks A, Brown EN et al.. (2006) OSI-930: a novel selective inhibitor of Kit and kinase insert domain receptor tyrosine kinases with antitumor activity in mouse xenograft models. Cancer Res, 66 (2): 1015-24. [PMID:16424037]
27. Gendreau SB, Ventura R, Keast P, Laird AD, Yakes FM, Zhang W, Bentzien F, Cancilla B, Lutman J, Chu F et al.. (2007) Inhibition of the T790M gatekeeper mutant of the epidermal growth factor receptor by EXEL-7647. Clin Cancer Res, 13 (12): 3713-23. [PMID:17575237]
28. Getmanova EV, Chen Y, Bloom L, Gokemeijer J, Shamah S, Warikoo V, Wang J, Ling V, Sun L. (2006) Antagonists to human and mouse vascular endothelial growth factor receptor 2 generated by directed protein evolution in vitro. Chem Biol, 13 (5): 549-56. [PMID:16720276]
29. Glaser KB, Li J, Marcotte PA, Magoc TJ, Guo J, Reuter DR, Tapang P, Wei RQ, Pease LJ, Bui MH et al.. (2012) Preclinical characterization of ABT-348, a kinase inhibitor targeting the aurora, vascular endothelial growth factor receptor/platelet-derived growth factor receptor, and Src kinase families. J Pharmacol Exp Ther, 343 (3): 617-27. [PMID:22935731]
30. Guagnano V, Furet P, Spanka C, Bordas V, Le Douget M, Stamm C, Brueggen J, Jensen MR, Schnell C, Schmid H et al.. (2011) Discovery of 3-(2,6-dichloro-3,5-dimethoxy-phenyl)-1-{6-[4-(4-ethyl-piperazin-1-yl)-phenylamino]-pyrimidin-4-yl}-1-methyl-urea (NVP-BGJ398), a potent and selective inhibitor of the fibroblast growth factor receptor family of receptor tyrosine kinase. J Med Chem, 54 (20): 7066-83. [PMID:21936542]
31. Harris PA, Boloor A, Cheung M, Kumar R, Crosby RM, Davis-Ward RG, Epperly AH, Hinkle KW, Hunter 3rd RN, Johnson JH et al.. (2008) Discovery of 5-[[4-[(2,3-dimethyl-2H-indazol-6-yl)methylamino]-2-pyrimidinyl]amino]-2-methyl-benzenesulfonamide (Pazopanib), a novel and potent vascular endothelial growth factor receptor inhibitor. J Med Chem, 51 (15): 4632-40. [PMID:18620382]
32. Harris PA, Cheung M, Hunter 3rd RN, Brown ML, Veal JM, Nolte RT, Wang L, Liu W, Crosby RM, Johnson JH et al.. (2005) Discovery and evaluation of 2-anilino-5-aryloxazoles as a novel class of VEGFR2 kinase inhibitors. J Med Chem, 48 (5): 1610-9. [PMID:15743202]
33. Hennequin LF, Thomas AP, Johnstone C, Stokes ES, Plé PA, Lohmann JJ, Ogilvie DJ, Dukes M, Wedge SR, Curwen JO et al.. (1999) Design and structure-activity relationship of a new class of potent VEGF receptor tyrosine kinase inhibitors. J Med Chem, 42 (26): 5369-89. [PMID:10639280]
34. Hilberg F, Roth GJ, Krssak M, Kautschitsch S, Sommergruber W, Tontsch-Grunt U, Garin-Chesa P, Bader G, Zoephel A, Quant J et al.. (2008) BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res, 68 (12): 4774-82. [PMID:18559524]
35. Hudkins RL, Becknell NC, Zulli AL, Underiner TL, Angeles TS, Aimone LD, Albom MS, Chang H, Miknyoczki SJ, Hunter K et al.. (2012) Synthesis and biological profile of the pan-vascular endothelial growth factor receptor/tyrosine kinase with immunoglobulin and epidermal growth factor-like homology domains 2 (VEGF-R/TIE-2) inhibitor 11-(2-methylpropyl)-12,13-dihydro-2-methyl-8-(pyrimidin-2-ylamino)-4H-indazolo[5,4-a]pyrrolo[3,4-c]carbazol-4-one (CEP-11981): a novel oncology therapeutic agent. J Med Chem, 55 (2): 903-13. [PMID:22148921]
36. Kammasud N, Boonyarat C, Sanphanya K, Utsintong M, Tsunoda S, Sakurai H, Saiki I, André I, Grierson DS, Vajragupta O. (2009) 5-Substituted pyrido[2,3-d]pyrimidine, an inhibitor against three receptor tyrosine kinases. Bioorg Med Chem Lett, 19 (3): 745-50. [PMID:19110422]
37. Khanwelkar RR, Chen GS, Wang HC, Yu CW, Huang CH, Lee O, Chen CH, Hwang CS, Ko CH, Chou NT et al.. (2010) Synthesis and structure-activity relationship of 6-arylureido-3-pyrrol-2-ylmethylideneindolin-2-one derivatives as potent receptor tyrosine kinase inhibitors. Bioorg Med Chem, 18 (13): 4674-86. [PMID:20570526]
38. Kim MH, Tsuhako AL, Co EW, Aftab DT, Bentzien F, Chen J, Cheng W, Engst S, Goon L, Klein RR et al.. (2012) The design, synthesis, and biological evaluation of potent receptor tyrosine kinase inhibitors. Bioorg Med Chem Lett, 22 (15): 4979-85. [PMID:22765894]
39. King C, Hristova K. (2019) Direct measurements of VEGF-VEGFR2 binding affinities reveal the coupling between ligand binding and receptor dimerization. J Biol Chem, 294 (23): 9064-9075. [PMID:31023826]
40. Kiselyov AS, Semenova M, Semenov VV, Piatnitski E, Ouyang S. (2006) Hetaryl imidazoles: a novel dual inhibitors of VEGF receptors I and II. Bioorg Med Chem Lett, 16 (5): 1440-4. [PMID:16321531]
41. Lam B, Arikawa Y, Cramlett J, Dong Q, de Jong R, Feher V, Grimshaw CE, Farrell PJ, Hoffman ID, Jennings A et al.. (2016) Discovery of TAK-659 an orally available investigational inhibitor of Spleen Tyrosine Kinase (SYK). Bioorg Med Chem Lett, 26 (24): 5947-5950. [PMID:27839918]
42. Lee K, Jeong KW, Lee Y, Song JY, Kim MS, Lee GS, Kim Y. (2010) Pharmacophore modeling and virtual screening studies for new VEGFR-2 kinase inhibitors. Eur J Med Chem, 45 (11): 5420-7. [PMID:20869793]
43. Li Y, Liu Y, Zhang D, Chen J, Yang G, Tang P, Yang C, Liu J, Zhang J, Ouyang L. (2023) Discovery, Synthesis, and Evaluation of Novel Dual Inhibitors of a Vascular Endothelial Growth Factor Receptor and Poly(ADP-Ribose) Polymerase for BRCA Wild-Type Breast Cancer Therapy. J Med Chem, 66 (17): 12069-12100. [PMID:37616488]
44. Liao AT, Chien MB, Shenoy N, Mendel DB, McMahon G, Cherrington JM, London CA. (2002) Inhibition of constitutively active forms of mutant kit by multitargeted indolinone tyrosine kinase inhibitors. Blood, 100 (2): 585-93. [PMID:12091352]
45. Lu D, Shen J, Vil MD, Zhang H, Jimenez X, Bohlen P, Witte L, Zhu Z. (2003) Tailoring in vitro selection for a picomolar affinity human antibody directed against vascular endothelial growth factor receptor 2 for enhanced neutralizing activity. J Biol Chem, 278 (44): 43496-507. [PMID:12917408]
46. Machrouhi F, Ouhamou N, Laderoute K, Calaoagan J, Bukhtiyarova M, Ehrlich PJ, Klon AE. (2010) The rational design of a novel potent analogue of the 5'-AMP-activated protein kinase inhibitor compound C with improved selectivity and cellular activity. Bioorg Med Chem Lett, 20 (22): 6394-9. [PMID:20932747]
47. Marathe P, Tang Y, Sleczka B, Rodrigues D, Gavai A, Wong T, Christopher L, Zhang H. (2010) Preclinical pharmacokinetics and in vitro metabolism of BMS-690514, a potent inhibitor of EGFR and VEGFR2. J Pharm Sci, 99 (8): 3579-93. [PMID:20166197]
48. McTigue M, Murray BW, Chen JH, Deng YL, Solowiej J, Kania RS. (2012) Molecular conformations, interactions, and properties associated with drug efficiency and clinical performance among VEGFR TK inhibitors. Proc Natl Acad Sci USA, 109 (45): 18281-9. [PMID:22988103]
49. McTigue MA, Wickersham JA, Pinko C, Showalter RE, Parast CV, Tempczyk-Russell A, Gehring MR, Mroczkowski B, Kan CC, Villafranca JE et al.. (1999) Crystal structure of the kinase domain of human vascular endothelial growth factor receptor 2: a key enzyme in angiogenesis. Structure, 7 (3): 319-30. [PMID:10368301]
50. Mendel DB, Laird AD, Xin X, Louie SG, Christensen JG, Li G, Schreck RE, Abrams TJ, Ngai TJ, Lee LB et al.. (2003) In vivo antitumor activity of SU11248, a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor and platelet-derived growth factor receptors: determination of a pharmacokinetic/pharmacodynamic relationship. Clin Cancer Res, 9 (1): 327-37. [PMID:12538485]
51. Musumeci F, Radi M, Brullo C, Schenone S. (2012) Vascular endothelial growth factor (VEGF) receptors: drugs and new inhibitors. J Med Chem, 55 (24): 10797-822. [PMID:23098265]
52. Nakagawa T, Tohyama O, Yamaguchi A, Matsushima T, Takahashi K, Funasaka S, Shirotori S, Asada M, Obaishi H. (2010) E7050: a dual c-Met and VEGFR-2 tyrosine kinase inhibitor promotes tumor regression and prolongs survival in mouse xenograft models. Cancer Sci, 101 (1): 210-5. [PMID:19832844]
53. Ohno H, Kubo K, Murooka H, Kobayashi Y, Nishitoba T, Shibuya M, Yoneda T, Isoe T. (2006) A c-fms tyrosine kinase inhibitor, Ki20227, suppresses osteoclast differentiation and osteolytic bone destruction in a bone metastasis model. Mol Cancer Ther, 5 (11): 2634-43. [PMID:17121910]
54. Pan BS, Chan GK, Chenard M, Chi A, Davis LJ, Deshmukh SV, Gibbs JB, Gil S, Hang G, Hatch H et al.. (2010) MK-2461, a novel multitargeted kinase inhibitor, preferentially inhibits the activated c-Met receptor. Cancer Res, 70 (4): 1524-33. [PMID:20145145]
55. Patwardhan PP, Ivy KS, Musi E, de Stanchina E, Schwartz GK. (2016) Significant blockade of multiple receptor tyrosine kinases by MGCD516 (Sitravatinib), a novel small molecule inhibitor, shows potent anti-tumor activity in preclinical models of sarcoma. Oncotarget, 7 (4): 4093-109. [PMID:26675259]
56. Patyna S, Laird AD, Mendel DB, O'farrell AM, Liang C, Guan H, Vojkovsky T, Vasile S, Wang X, Chen J et al.. (2006) SU14813: a novel multiple receptor tyrosine kinase inhibitor with potent antiangiogenic and antitumor activity. Mol Cancer Ther, 5 (7): 1774-82. [PMID:16891463]
57. Pollard JR, Mortimore M. (2009) Discovery and development of aurora kinase inhibitors as anticancer agents. J Med Chem, 52 (9): 2629-51. [PMID:19320489]
58. Qi B, Yang Y, Gong G, He H, Yue X, Xu X, Hu Y, Li J, Chen T, Wan X et al.. (2019) Discovery of N1-(4-((7-(3-(4-ethylpiperazin-1-yl)propoxy)-6-methoxyquinolin-4-yl)oxy)-3,5-difluorophenyl)-N3-(2-(2,6-difluorophenyl)-4-oxothiazolidin-3-yl)urea as a multi-tyrosine kinase inhibitor for drug-sensitive and drug-resistant cancers treatment. Eur J Med Chem, 163: 10-27. [PMID:30503936]
59. Renhowe PA, Pecchi S, Shafer CM, Machajewski TD, Jazan EM, Taylor C, Antonios-McCrea W, McBride CM, Frazier K, Wiesmann M et al.. (2009) Design, structure-activity relationships and in vivo characterization of 4-amino-3-benzimidazol-2-ylhydroquinolin-2-ones: a novel class of receptor tyrosine kinase inhibitors. J Med Chem, 52 (2): 278-92. [PMID:19113866]
60. Sammond DM, Nailor KE, Veal JM, Nolte RT, Wang L, Knick VB, Rudolph SK, Truesdale AT, Nartey EN, Stafford JA et al.. (2005) Discovery of a novel and potent series of dianilinopyrimidineurea and urea isostere inhibitors of VEGFR2 tyrosine kinase. Bioorg Med Chem Lett, 15 (15): 3519-23. [PMID:15990302]
61. Saxty G, Murray CW, Berdini V, Besong GE, Hamlett CCF, Johnson CN, Woodhead SJ, Reader M, Rees DC, Mevellec LA et al.. (2011) Pyrazolyl quinazoline kinase inhibitors. Patent number: WO2011135376 A1. Assignee: Astex Therapeutics Limited. Priority date: 30/04/2010. Publication date: 03/11/2011.
62. Su W-G, Zhang W, Jia H, Cui Y, Ren Y, Sai Y, Wu Z, Li W, Shao J. (2011) Compound, certain novel forms thereof, pharmaceutical compositions thereof and methods for preparation and use. Patent number: WO2011060746. Assignee: Hutchison Medipharma Limited. Priority date: 23/11/2009. Publication date: 26/05/2011.
63. Su Y, Wang J, Bao R. (2020) Inhibitor containing bicyclic derivative, preparation method therefor and use thereof. Patent number: WO2020228756A1. Assignee: Shanghai Hansoh Biomedical Technology Co., Ltd., Jiangsu Hansoh Pharmaceutical Group Co., Ltd.. Priority date: 14/05/2020. Publication date: 19/11/2020.
64. Subbiah V, Gainor JF, Rahal R, Brubaker JD, Kim JL, Maynard M, Hu W, Cao Q, Sheets MP, Wilson D et al.. (2018) Precision Targeted Therapy with BLU-667 for RET-Driven Cancers. Cancer Discov, 8 (7): 836-849. [PMID:29657135]
65. Suh B-C, Salgaonkar PD, Lee J, Koh JS, Song H-J, Lee IY, Lee J, Jung DS, Kim J-H, Kim S-W. (2016) Compounds and compositions for modulating EGFR mutant kinase activities. Patent number: WO2016060443A2. Assignee: Yuhan Corporation. Priority date: 13/10/2014. Publication date: 21/04/2016.
66. Sun L, Liang C, Shirazian S, Zhou Y, Miller T, Cui J, Fukuda JY, Chu JY, Nematalla A, Wang X et al.. (2003) Discovery of 5-[5-fluoro-2-oxo-1,2- dihydroindol-(3Z)-ylidenemethyl]-2,4- dimethyl-1H-pyrrole-3-carboxylic acid (2-diethylaminoethyl)amide, a novel tyrosine kinase inhibitor targeting vascular endothelial and platelet-derived growth factor receptor tyrosine kinase. J Med Chem, 46 (7): 1116-9. [PMID:12646019]
67. Sun L, Tran N, Liang C, Hubbard S, Tang F, Lipson K, Schreck R, Zhou Y, McMahon G, Tang C. (2000) Identification of substituted 3-[(4,5,6, 7-tetrahydro-1H-indol-2-yl)methylene]-1,3-dihydroindol-2-ones as growth factor receptor inhibitors for VEGF-R2 (Flk-1/KDR), FGF-R1, and PDGF-Rbeta tyrosine kinases. J Med Chem, 43 (14): 2655-63. [PMID:10893303]
68. Sun L, Tran N, Liang C, Tang F, Rice A, Schreck R, Waltz K, Shawver LK, McMahon G, Tang C. (1999) Design, synthesis, and evaluations of substituted 3-[(3- or 4-carboxyethylpyrrol-2-yl)methylidenyl]indolin-2-ones as inhibitors of VEGF, FGF, and PDGF receptor tyrosine kinases. J Med Chem, 42 (25): 5120-30. [PMID:10602697]
69. Sun L, Tran N, Tang F, App H, Hirth P, McMahon G, Tang C. (1998) Synthesis and biological evaluations of 3-substituted indolin-2-ones: a novel class of tyrosine kinase inhibitors that exhibit selectivity toward particular receptor tyrosine kinases. J Med Chem, 41 (14): 2588-603. [PMID:9651163]
70. Sun Q, Zhou J, Zhang Z, Guo M, Liang J, Zhou F, Long J, Zhang W, Yin F, Cai H et al.. (2014) Discovery of fruquintinib, a potent and highly selective small molecule inhibitor of VEGFR 1, 2, 3 tyrosine kinases for cancer therapy. Cancer Biol Ther, 15 (12): 1635-45. [PMID:25482937]
71. Sun X, Wang L, Yang H, Hu H. (2020) Crystal forms of compound, preparation method therefor, pharmaceutical composition and application thereof. Patent number: WO2020216188. Assignee: Beijing Kangchen Pharmaceutical Co., Ltd.. Priority date: 20/04/2020. Publication date: 29/10/2020.
72. Tap WD, Wainberg ZA, Anthony SP, Ibrahim PN, Zhang C, Healey JH, Chmielowski B, Staddon AP, Cohn AL, Shapiro GI et al.. (2015) Structure-Guided Blockade of CSF1R Kinase in Tenosynovial Giant-Cell Tumor. N Engl J Med, 373 (5): 428-37. [PMID:26222558]
73. Tian S, Quan H, Xie C, Guo H, Lü F, Xu Y, Li J, Lou L. (2011) YN968D1 is a novel and selective inhibitor of vascular endothelial growth factor receptor-2 tyrosine kinase with potent activity in vitro and in vivo. Cancer Sci, 102 (7): 1374-80. [PMID:21443688]
74. Tovar C, Higgins B, Deo D, Kolinsky K, Liu JJ, Heimbrook DC, Vassilev LT. (2010) Small-molecule inducer of cancer cell polyploidy promotes apoptosis or senescence: Implications for therapy. Cell Cycle, 9 (16): 3364-75. [PMID:20814247]
75. Traxler P, Allegrini PR, Brandt R, Brueggen J, Cozens R, Fabbro D, Grosios K, Lane HA, McSheehy P, Mestan J et al.. (2004) AEE788: a dual family epidermal growth factor receptor/ErbB2 and vascular endothelial growth factor receptor tyrosine kinase inhibitor with antitumor and antiangiogenic activity. Cancer Res, 64 (14): 4931-41. [PMID:15256466]
76. Trudel S, Li ZH, Wei E, Wiesmann M, Chang H, Chen C, Reece D, Heise C, Stewart AK. (2005) CHIR-258, a novel, multitargeted tyrosine kinase inhibitor for the potential treatment of t(4;14) multiple myeloma. Blood, 105 (7): 2941-8. [PMID:15598814]
77. Vieth M, Cummins DJ. (2000) DoMCoSAR: a novel approach for establishing the docking mode that is consistent with the structure-activity relationship. Application to HIV-1 protease inhibitors and VEGF receptor tyrosine kinase inhibitors. J Med Chem, 43 (16): 3020-32. [PMID:10956210]
78. Wang D, Tang F, Wang S, Jiang Z, Zhang L. (2012) Preclinical anti-angiogenesis and anti-tumor activity of SIM010603, an oral, multi-targets receptor tyrosine kinases inhibitor. Cancer Chemother Pharmacol, 69 (1): 173-83. [PMID:21638122]
79. Wedge SR, Kendrew J, Hennequin LF, Valentine PJ, Barry ST, Brave SR, Smith NR, James NH, Dukes M, Curwen JO et al.. (2005) AZD2171: a highly potent, orally bioavailable, vascular endothelial growth factor receptor-2 tyrosine kinase inhibitor for the treatment of cancer. Cancer Res, 65 (10): 4389-400. [PMID:15899831]
80. Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M et al.. (2004) BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res, 64 (19): 7099-109. [PMID:15466206]
81. Wilhelm SM, Dumas J, Adnane L, Lynch M, Carter CA, Schütz G, Thierauch KH, Zopf D. (2011) Regorafenib (BAY 73-4506): a new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int J Cancer, 129 (1): 245-55. [PMID:21170960]
82. Wodicka LM, Ciceri P, Davis MI, Hunt JP, Floyd M, Salerno S, Hua XH, Ford JM, Armstrong RC, Zarrinkar PP et al.. (2010) Activation state-dependent binding of small molecule kinase inhibitors: structural insights from biochemistry. Chem Biol, 17 (11): 1241-9. [PMID:21095574]
83. Wu F. (2021) Multi-kinase inhibitor compound, and crystal form and use thereof. Patent number: US10889586B2. Assignee: Nanjing Transthera Biosciences Co Ltd. Priority date: 13/12/2016. Publication date: 12/01/2021.
84. Xi N. (2015) Substituted quinoline compounds and methods of use. Patent number: US9133162B2. Assignee: Sunshine Lake Pharma Co Ltd, Calitor Sciences LLC. Priority date: 28/02/2011. Publication date: 15/09/2015.
85. Yakes FM, Chen J, Tan J, Yamaguchi K, Shi Y, Yu P, Qian F, Chu F, Bentzien F, Cancilla B et al.. (2011) Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol Cancer Ther, 10 (12): 2298-308. [PMID:21926191]
86. You WK, Sennino B, Williamson CW, Falcón B, Hashizume H, Yao LC, Aftab DT, McDonald DM. (2011) VEGF and c-Met blockade amplify angiogenesis inhibition in pancreatic islet cancer. Cancer Res, 71 (14): 4758-68. [PMID:21613405]
87. Zhao G, Li WY, Chen D, Henry JR, Li HY, Chen Z, Zia-Ebrahimi M, Bloem L, Zhai Y, Huss K et al.. (2011) A novel, selective inhibitor of fibroblast growth factor receptors that shows a potent broad spectrum of antitumor activity in several tumor xenograft models. Mol Cancer Ther, 10 (11): 2200-10. [PMID:21900693]
88. Zhou Y, Chen Y, Tong L, Xie H, Wen W, Zhang J, Xi Y, Shen Y, Geng M, Wang Y et al.. (2012) AL3810, a multi-tyrosine kinase inhibitor, exhibits potent anti-angiogenic and anti-tumour activity via targeting VEGFR, FGFR and PDGFR. J Cell Mol Med, 16 (10): 2321-30. [PMID:22304225]
89. Zhou Y, Shan S, Li ZB, Xin LJ, Pan DS, Yang QJ, Liu YP, Yue XP, Liu XR, Gao JZ et al.. (2017) CS2164, a novel multi-target inhibitor against tumor angiogenesis, mitosis and chronic inflammation with anti-tumor potency. Cancer Sci, 108 (3): 469-477. [PMID:28004478]
How to cite this page
Type IV RTKs: VEGF (vascular endothelial growth factor) receptor family: kinase insert domain receptor. Last modified on 28/08/2023. Accessed on 26/04/2024. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetoimmunopharmacology.org/GRAC/ObjectDisplayForward?objectId=1813.