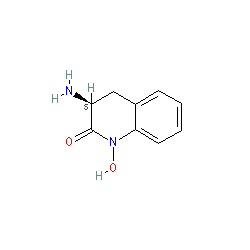
|
Synonyms: compound 1 [PMID: 23466229]
Compound class:
Synthetic organic
Comment: PF-04859989 was identified as a potent, irreversable and selective inhibitor of human and rat kynurenine aminotransferase II (KATII; a.k.a. aminoadipate aminotransferase AADAT) [1-2] . An X-ray crystal structure of the enzyme co-crystalised with an inhibitor has been reported [1]. In vivo pharmacokinetic and efficacy studies in rat show it to be brain-penetrant, reducing brain kynurenic acid by 50%. KAT inhibitors are expected to exhibit neuroprotective effects, that may be beneficial for the treatment of neurodegenerative and cognitive impairment disorders [3].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Dounay AB, Anderson M, Bechle BM, Campbell BM, Claffey MM, Evdokimov A, Evrard E, Fonseca KR, Gan X, Ghosh S et al.. (2012)
Discovery of Brain-Penetrant, Irreversible Kynurenine Aminotransferase II Inhibitors for Schizophrenia. ACS Med Chem Lett, 3 (3): 187-92. [PMID:24900455] |
|
2. Dounay AB, Anderson M, Bechle BM, Evrard E, Gan X, Kim JY, McAllister LA, Pandit J, Rong S, Salafia MA et al.. (2013)
PF-04859989 as a template for structure-based drug design: identification of new pyrazole series of irreversible KAT II inhibitors with improved lipophilic efficiency. Bioorg Med Chem Lett, 23 (7): 1961-6. [PMID:23466229] |
|
3. Nematollahi A, Sun G, Jayawickrama GS, Church WB. (2016)
Kynurenine Aminotransferase Isozyme Inhibitors: A Review. Int J Mol Sci, 17 (6). [PMID:27314340] |
|
4. Nguyen NT, Kimura A, Nakahama T, Chinen I, Masuda K, Nohara K, Fujii-Kuriyama Y, Kishimoto T. (2010)
Aryl hydrocarbon receptor negatively regulates dendritic cell immunogenicity via a kynurenine-dependent mechanism. Proc Natl Acad Sci USA, 107 (46): 19961-6. [PMID:21041655] |
|
5. Wang Y, Liu H, McKenzie G, Witting PK, Stasch JP, Hahn M, Changsirivathanathamrong D, Wu BJ, Ball HJ, Thomas SR et al.. (2010)
Kynurenine is an endothelium-derived relaxing factor produced during inflammation. Nat Med, 16 (3): 279-85. [PMID:20190767] |