|
Compound class:
Synthetic organic
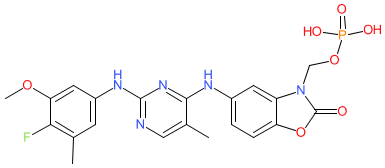
Comment: Fosifidancitinib is a Janus kinase (JAK) inhibitor that is claimed in Rigel Pharmaceuticals' patent WO2010085684A1 [1]. Strictly I-538 refers to the sodium salt but we represent the parent molecule in this entry. The chemical structure that was submitted to the WHO for the INN fosifidancitinib matches the structure of example I-538 (minus the sodium). Rigel declare two JAK inhibitor programmes on their pipeline webpage; one in partnership with Aclaris Therapeutics (ATI-501/ATI-502, oral and topical formulations respectively) for dermatological applications, and the second in partnership with AstraZeneca through which AZD0449 (R256) is being investigated as an inhalation asthma therapeutic. None of these investigational agents have published name to structure declarations.
|
|
|||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| JAK2/3 inhibitors have clinical application in the treatment of inflammatory conditions via downregulation of pathological JAK-STAT pathway activation. In particular JAK3 appears to associate with the common gamma chain (γc) of various cytokine receptors and is therefore important for the cellular responses to a wide range of cytokines including IL-2, IL-4, IL-7, IL-9, IL-15 and IL-21. |