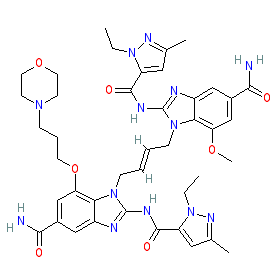
|
Synonyms: compound 3 [PMID: 30405246] | diABZI | diABZI STING agonist-1
Compound class:
Synthetic organic
Comment: This compound is an optimised derivative of STING agonist 2 that has improved solubility and improved potency in primary cells [4]. Like agonist 2 it is one of the chemical structures claimed in GlaxoSmithKline's patent WO2017175147A1 [1]. it behaves as a cyclic dinucleotide mimic to activate the STING pathway.
SARS-CoV-2: Preclinical experimental evidence (in mice) suggests that this STING agonist may be effective in controlling infection by a range of SARS-CoV-2 variants, including B.1.351 (the beta variant [3]) [2]. The mechanistic hypothesis is that directly activating STING transiently stimulates interferon (IFN) signalling and circumvents the virus' ability to block IFN activation in infected respiratory epithelial cells, and re-establishes the innate immune response to infection. |
|
|||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| Agonist 3 induces IFNβ production in human PBMCs with an EC50 of 130 nM (compared to the EC50 of agonist 2 which is 3100 nM) [4]. It promotes an adaptive CD8+ T cell response in vivo that manifests as a significant reduction in tumour volume (even complete tumour regression) when delivered subcutaneously in mice with CT-26 colorectal tumour xenografts. This indicates the systemic activity of the compound as an anti-tumour immune-modulating agent. Agonist 3 induces production of IL-6, TNF, and CXCL1 in wild-type but not Sting knockout mice, which indicates the Sting-dependency of its effect. |