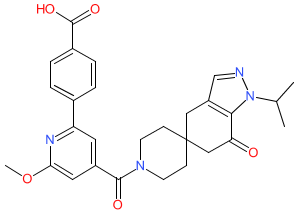
|
Synonyms: PF-05221304 | PF05221304
Compound class:
Synthetic organic
Comment: Clesacostat (PF-05221304) is an acetyl-CoA carboxylase (ACC) inhibitor [3]. It was designed to treat non-alcoholic steatohepatitis (NASH) by targeting disordered lipid metabolism in the liver.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Clesacostat (PF-05221304) has progressed to clinical evaluation. It is being investigated as a monotherpay and in combination with the DGAT2 inhibitor ervogastat. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT04321031 | Metabolic Interventions to Resolve Non-alcoholic Steatohepatitis (NASH) With Fibrosis (MIRNA) | Phase 2 Interventional | Pfizer | 1 | |
| NCT03776175 | A Study To Assess Pharmacodynamics, Safety And Tolerability Of PF-05221304 And PF-06865571 Co-Administered For 6 Weeks In Adults With Non-Alcoholic Fatty Liver Disease. | Phase 2 Interventional | Pfizer | 2 | |
| NCT04399538 | Study of Pharmacodynamics and Safety of DGAT2i and ACCi Coadministered in Participants With Sponsor-defined Presumed Non Alcoholic Steatohepatitis | Phase 2 Interventional | Pfizer | ||