|
Synonyms: compound 21 [WO2021053495A1]
Compound class:
Synthetic organic
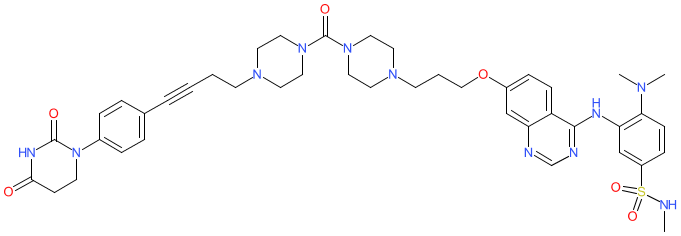
Comment: This compound is a bifunctional degrader of the cardiac-specific kinase TNNI3K protein, that is claimed in patent WO2021053495A1 [1]. It's kinase binding domain is derived from the quinazoline TNNI3K inhibitor GSK114 [2], and it contains a cereblon binding ligand to promote association with the protein-degrading E3 ubiquitin ligase complex.
|
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Arista L, Broennimann V, D'Alessandro PL, Doumampoum-Metoul L, Goude M-L, Hebach C, Hollingworth GJ, Jeulin IKJ, Kirman LC, Lorber J et al.. (2021)
Bifunctional degraders and their methods of use. Patent number: WO2021053495A1. Assignee: Novartis Ag. Priority date: 16/09/2019. Publication date: 25/03/2021. |
|
2. Lawhorn BG, Philp J, Graves AP, Shewchuk L, Holt DA, Gatto Jr GJ, Kallander LS. (2016)
GSK114: A selective inhibitor for elucidating the biological role of TNNI3K. Bioorg Med Chem Lett, 26 (14): 3355-3358. [PMID:27246618] |