|
Synonyms: peptide 3c [PMID: 31611414]
Compound class:
Synthetic organic
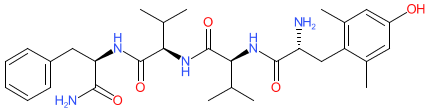
Comment: Bilorphin is a derivative of a naturally ocurring tetrapeptide that was isolated from an Australian estuarine isolate of Penicillium species MST-MF667 [1]. It acts as a G protein signalling biased agonist of the μ-opioid receptor in vitro. Bilorphin promotes minimal β-arrestin recruitment and receptor internalization. Systemic and oral activity was achieved by glycosylating the C-terminal residue of bilorphin to create bilactorphin. The in vivo analgesic potency of bilactorphin is similar to that of morphine.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| Selectivity at GPCRs | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||