GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
mitogen-activated protein kinase 11
 Target has curated data in GtoImmuPdb
Target has curated data in GtoImmuPdb
Target id: 1500
Nomenclature: mitogen-activated protein kinase 11
Abbreviated Name: p38β
Family: p38 subfamily
Contents:
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 364 | 22q13.33 | MAPK11 | mitogen-activated protein kinase 11 | |
| Mouse | - | 364 | 15 E3 | Mapk11 | mitogen-activated protein kinase 11 | |
| Rat | - | 364 | 7q34 | Mapk11 | mitogen-activated protein kinase 11 | |
Previous and Unofficial Names  |
| SAPK2 | p38Beta | PRKM11 |
Database Links  |
|
| Alphafold | Q15759 (Hs), Q9WUI1 (Mm) |
| BRENDA | 2.7.11.24 |
| ChEMBL Target | CHEMBL3961 (Hs), CHEMBL4335 (Mm) |
| DrugBank Target | Q15759 (Hs) |
| Ensembl Gene | ENSG00000185386 (Hs), ENSMUSG00000053137 (Mm), ENSRNOG00000006984 (Rn) |
| Entrez Gene | 5600 (Hs), 19094 (Mm), 689314 (Rn) |
| Human Protein Atlas | ENSG00000185386 (Hs) |
| KEGG Enzyme | 2.7.11.24 |
| KEGG Gene | hsa:5600 (Hs), mmu:19094 (Mm), rno:689314 (Rn) |
| OMIM | 602898 (Hs) |
| Pharos | Q15759 (Hs) |
| RefSeq Nucleotide | NM_002751 (Hs), NM_011161 (Mm), NM_001109532 (Rn) |
| RefSeq Protein | NP_002742 (Hs), NP_035291 (Mm), NP_001103002 (Rn) |
| UniProtKB | Q15759 (Hs), Q9WUI1 (Mm) |
| Wikipedia | MAPK11 (Hs) |
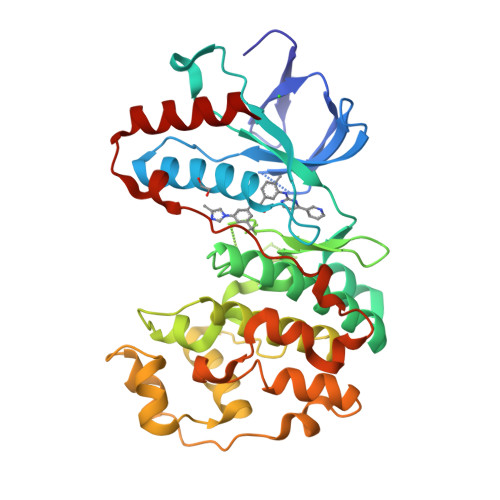
Selected 3D Structures  |
|||||||||||||
|
|
||||||||||||
Enzyme Reaction  |
||||
|
||||
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
DiscoveRx KINOMEscan® screen  |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A screen of 72 inhibitors against 456 human kinases. Quantitative data were derived using DiscoveRx KINOMEscan® platform. http://www.discoverx.com/services/drug-discovery-development-services/kinase-profiling/kinomescan Reference: 5,21 |
 
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Target used in screen: p38-beta | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displaying the top 10 most potent ligands View all ligands in screen » | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
EMD Millipore KinaseProfilerTM screen/Reaction Biology Kinase HotspotSM screen  |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
A screen profiling 158 kinase inhibitors (Calbiochem Protein Kinase Inhibitor Library I and II, catalogue numbers 539744 and 539745) for their inhibitory activity at 1µM and 10µM against 234 human recombinant kinases using the EMD Millipore KinaseProfilerTM service. A screen profiling the inhibitory activity of 178 commercially available kinase inhibitors at 0.5µM against a panel of 300 recombinant protein kinases using the Reaction Biology Corporation Kinase HotspotSM platform. http://www.millipore.com/techpublications/tech1/pf3036 http://www.reactionbiology.com/webapps/main/pages/kinase.aspx Reference: 1,8 |
 
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Target used in screen: SAPK2b/P38b(MAPK11) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displaying the top 10 most potent ligands View all ligands in screen » | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| p38 MAP kinases are ubiquitous, highly conserved enzymes which regulate the production of proinflammatory mediators (such as TNFα and IL-1) in response to inflammatory cytokines or environmental stress [11-12,14,17-18,20]. They are essential for normal immune and inflammatory responses, but are also involved in many other cellular processes such as regulating the cell cycle and cytoskeletal remodelling. Pharmacological inhibition of p38 MAP kinases reduces inflammatory cytokine synthesis, making these enzymes validated and extensively pursued drug targets for autoimmune and inflammatory diseases, including arthritis and other joint diseases, septic shock, myocardial injury and neuroinflammation. A number of pan-p38 MAP kinase inhibitors and isoform selective inhibitors have been evaluated in clinical trials, although none have yet reached the clinic. |
References
1. Anastassiadis T, Deacon SW, Devarajan K, Ma H, Peterson JR. (2011) Comprehensive assay of kinase catalytic activity reveals features of kinase inhibitor selectivity. Nat Biotechnol, 29 (11): 1039-45. [PMID:22037377]
2. Armani E, Capaldi C, Bagnacani V, Saccani F, Aquino G, Puccini P, Facchinetti F, Martucci C, Moretto N, Villetti G et al.. (2022) Design, Synthesis, and Biological Characterization of Inhaled p38α/β MAPK Inhibitors for the Treatment of Lung Inflammatory Diseases. J Med Chem, 65 (10): 7170-7192. [PMID:35546685]
3. Bachegowda L, Morrone K, Winski SL, Mantzaris I, Bartenstein M, Ramachandra N, Giricz O, Sukrithan V, Nwankwo G, Shahnaz S et al.. (2016) Pexmetinib: A Novel Dual Inhibitor of Tie2 and p38 MAPK with Efficacy in Preclinical Models of Myelodysplastic Syndromes and Acute Myeloid Leukemia. Cancer Res, 76 (16): 4841-4849. [PMID:27287719]
4. Bhattacharjee D, Bakar J, Chitnis SP, Sausville EL, Ashtekar KD, Mendelson BE, Long K, Smith JC, Heppner DE, Sheltzer JM. (2023) Inhibition of a lower potency target drives the anticancer activity of a clinical p38 inhibitor. Cell Chem Biol, 30 (10): 1211-1222.e5. [PMID:37827156]
5. Davis MI, Hunt JP, Herrgard S, Ciceri P, Wodicka LM, Pallares G, Hocker M, Treiber DK, Zarrinkar PP. (2011) Comprehensive analysis of kinase inhibitor selectivity. Nat Biotechnol, 29 (11): 1046-51. [PMID:22037378]
6. Devadas B, Selness SR, Xing L, Madsen HM, Marrufo LD, Shieh H, Messing DM, Yang JZ, Morgan HM, Anderson GD et al.. (2011) Substituted N-aryl-6-pyrimidinones: a new class of potent, selective, and orally active p38 MAP kinase inhibitors. Bioorg Med Chem Lett, 21 (13): 3856-60. [PMID:21620699]
7. Eyers PA, Craxton M, Morrice N, Cohen P, Goedert M. (1998) Conversion of SB 203580-insensitive MAP kinase family members to drug-sensitive forms by a single amino-acid substitution. Chem Biol, 5 (6): 321-8. [PMID:9653550]
8. Gao Y, Davies SP, Augustin M, Woodward A, Patel UA, Kovelman R, Harvey KJ. (2013) A broad activity screen in support of a chemogenomic map for kinase signalling research and drug discovery. Biochem J, 451 (2): 313-28. [PMID:23398362]
9. Goldstein DM, Kuglstatter A, Lou Y, Soth MJ. (2010) Selective p38alpha inhibitors clinically evaluated for the treatment of chronic inflammatory disorders. J Med Chem, 53 (6): 2345-53. [PMID:19950901]
10. Goldstein DM, Soth M, Gabriel T, Dewdney N, Kuglstatter A, Arzeno H, Chen J, Bingenheimer W, Dalrymple SA, Dunn J et al.. (2011) Discovery of 6-(2,4-difluorophenoxy)-2-[3-hydroxy-1-(2-hydroxyethyl)propylamino]-8-methyl-8H-pyrido[2,3-d]pyrimidin-7-one (pamapimod) and 6-(2,4-difluorophenoxy)-8-methyl-2-(tetrahydro-2H-pyran-4-ylamino)pyrido[2,3-d]pyrimidin-7(8H)-one (R1487) as orally bioavailable and highly selective inhibitors of p38α mitogen-activated protein kinase. J Med Chem, 54 (7): 2255-65. [PMID:21375264]
11. Han J, Jiang Y, Li Z, Kravchenko VV, Ulevitch RJ. (1997) Activation of the transcription factor MEF2C by the MAP kinase p38 in inflammation. Nature, 386 (6622): 296-9. [PMID:9069290]
12. Han J, Lee JD, Bibbs L, Ulevitch RJ. (1994) A MAP kinase targeted by endotoxin and hyperosmolarity in mammalian cells. Science, 265 (5173): 808-11. [PMID:7914033]
13. Karaman MW, Herrgard S, Treiber DK, Gallant P, Atteridge CE, Campbell BT, Chan KW, Ciceri P, Davis MI, Edeen PT et al.. (2008) A quantitative analysis of kinase inhibitor selectivity. Nat Biotechnol, 26 (1): 127-32. [PMID:18183025]
14. Lee JC, Kumar S, Griswold DE, Underwood DC, Votta BJ, Adams JL. (2000) Inhibition of p38 MAP kinase as a therapeutic strategy. Immunopharmacology, 47 (2-3): 185-201. [PMID:10878289]
15. Miwatashi S, Arikawa Y, Kotani E, Miyamoto M, Naruo K, Kimura H, Tanaka T, Asahi S, Ohkawa S. (2005) Novel inhibitor of p38 MAP kinase as an anti-TNF-alpha drug: discovery of N-[4-[2-ethyl-4-(3-methylphenyl)-1,3-thiazol-5-yl]-2-pyridyl]benzamide (TAK-715) as a potent and orally active anti-rheumatoid arthritis agent. J Med Chem, 48 (19): 5966-79. [PMID:16162000]
16. Moffett K, Konteatis Z, Nguyen D, Shetty R, Ludington J, Fujimoto T, Lee KJ, Chai X, Namboodiri H, Karpusas M et al.. (2011) Discovery of a novel class of non-ATP site DFG-out state p38 inhibitors utilizing computationally assisted virtual fragment-based drug design (vFBDD). Bioorg Med Chem Lett, 21 (23): 7155-65. [PMID:22014550]
17. Pearson G, Robinson F, Beers Gibson T, Xu BE, Karandikar M, Berman K, Cobb MH. (2001) Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr Rev, 22 (2): 153-83. [PMID:11294822]
18. Raingeaud J, Gupta S, Rogers JS, Dickens M, Han J, Ulevitch RJ, Davis RJ. (1995) Pro-inflammatory cytokines and environmental stress cause p38 mitogen-activated protein kinase activation by dual phosphorylation on tyrosine and threonine. J Biol Chem, 270 (13): 7420-6. [PMID:7535770]
19. Tan X, Tester RW, Luedtke GR, Chakravarty S, Mavunkel BJ, Perumattam JJ, Lu Q, Nashashibi I, Jung J, Hu J et al.. (2010) Design and synthesis of piperazine-indole p38 alpha MAP kinase inhibitors with improved pharmacokinetic profiles. Bioorg Med Chem Lett, 20 (3): 828-31. [PMID:20071169]
20. Wang XZ, Ron D. (1996) Stress-induced phosphorylation and activation of the transcription factor CHOP (GADD153) by p38 MAP Kinase. Science, 272 (5266): 1347-9. [PMID:8650547]
21. Wodicka LM, Ciceri P, Davis MI, Hunt JP, Floyd M, Salerno S, Hua XH, Ford JM, Armstrong RC, Zarrinkar PP et al.. (2010) Activation state-dependent binding of small molecule kinase inhibitors: structural insights from biochemistry. Chem Biol, 17 (11): 1241-9. [PMID:21095574]
How to cite this page
p38 subfamily: mitogen-activated protein kinase 11. Last modified on 13/10/2023. Accessed on 04/05/2026. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetoimmunopharmacology.org/GRAC/ObjectDisplayForward?objectId=1500.