|
Synonyms: DCC-2618 | DCC2618 | Quinlock®
ripretinib is an approved drug (FDA (2020), EMA (2021))
Compound class:
Synthetic organic
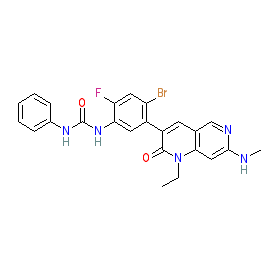
Comment: Ripretinib (DCC-2618) is an orally bioavailable, potent pan-KIT and PDGFRα kinase inhibitor that was developed by Dicephera Pharmaceuticals. It was designed for the treatment of malignancies that are driven by KIT and/or PDGFRα, for example gastrointestinal stromal tumors, glioblastoma multiforme and systemic mastocytosis. Ripretinib has activity across a broad range of resistance mutations in its target kinases, that emerge in response to imatinib treatment. Ripretinib binds to the 'switch pocket' of KIT, a domain that regulates the enzyme's catalytic conformation. Compounds that act in this way are often referred to as switch control inhibitors [1]. Another example of a switch control inhibitor is avapritinib.
The chemical structure shown here matches that supplied to the WHO for the INN, and this is claimed in patent WO2010051373 [2] as Example 39. Note that some PubChem SIDs are linked to an incorrect structure (as represented in PubChem CID 46208890). |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Bai Y, Bandara G, Ching Chan E, Maric I, Simakova O, Bandara SN, Lu WP, Wise SC, Flynn DL, Metcalfe DD et al.. (2013)
Targeting the KIT activating switch control pocket: a novel mechanism to inhibit neoplastic mast cell proliferation and mast cell activation. Leukemia, 27 (2): 278-85. [PMID:22907049] |
|
2. Flynn DL, Petillo PA, Kaufman MD. (2010)
Cyclopropane amides and analogs exhibiting anti-cancer and anti-proliferative activities. Patent number: WO2010051373. Assignee: Deciphera Pharmaceuticals, Llc. Priority date: 29/10/2008. Publication date: 06/05/2010. |