|
Synonyms: DCC-2618 | DCC2618 | Quinlock®
ripretinib is an approved drug (FDA (2020), EMA (2021))
Compound class:
Synthetic organic
Comment: Ripretinib (DCC-2618) is an orally bioavailable, potent pan-KIT and PDGFRα kinase inhibitor that was developed by Dicephera Pharmaceuticals. It was designed for the treatment of malignancies that are driven by KIT and/or PDGFRα, for example gastrointestinal stromal tumors, glioblastoma multiforme and systemic mastocytosis. Ripretinib has activity across a broad range of resistance mutations in its target kinases, that emerge in response to imatinib treatment. Ripretinib binds to the 'switch pocket' of KIT, a domain that regulates the enzyme's catalytic conformation. Compounds that act in this way are often referred to as switch control inhibitors [1]. Another example of a switch control inhibitor is avapritinib.
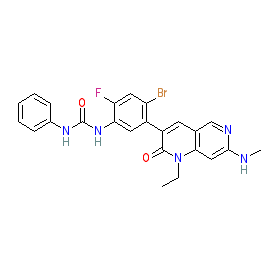
The chemical structure shown here matches that supplied to the WHO for the INN, and this is claimed in patent WO2010051373 [2] as Example 39. Note that some PubChem SIDs are linked to an incorrect structure (as represented in PubChem CID 46208890). |
|
|||||||||||||||||||||||||||||||||||
| Bioactivity Comments |
| DCC-2618 inhibits mutant forms of KIT which are responsible for the development of resistance to current therapeutics, including mutants found in primary and treatment-resistant GIST. It also inhibits the KIT D816V mutation which drives development of some cases of mast cell neoplasms (mastocytosis). Patent WO2010051373 [2] contains preclinical evaluation of compounds claimed therein, but only provides 'binned' indications of binding affinities to target kinases, not precise IC50 values. |
| Selectivity at catalytic receptors | ||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | |||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||