GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
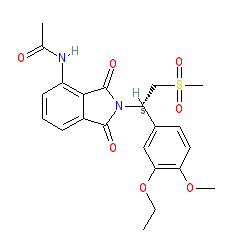
Synonyms: CC-10004 | Otezla®
apremilast is an approved drug (FDA (2014), EMA (2015))
Compound class:
Synthetic organic
Comment: Apremilast is an oral small molecule phosphodiesterase 4 (PDE4) inhibitor [1-2] (additional comment in [4]). It is selective for PDE4 over other PDE isozymes, but non-selective over PDE4 isoforms [5].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖
View more information in the IUPHAR Pharmacology Education Project: apremilast |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Gudjonsson JE, Johnston A, Ellis CN. (2012)
Novel systemic drugs under investigation for the treatment of psoriasis. J Am Acad Dermatol, 67 (1): 139-47. [PMID:22305044] |
|
2. Man HW, Schafer P, Wong LM, Patterson RT, Corral LG, Raymon H, Blease K, Leisten J, Shirley MA, Tang Y et al.. (2009)
Discovery of (S)-N-[2-[1-(3-ethoxy-4-methoxyphenyl)-2-methanesulfonylethyl]-1,3-dioxo-2,3-dihydro-1H-isoindol-4-yl] acetamide (apremilast), a potent and orally active phosphodiesterase 4 and tumor necrosis factor-alpha inhibitor. J Med Chem, 52 (6): 1522-4. [PMID:19256507] |
|
3. Schafer P. (2012)
Apremilast mechanism of action and application to psoriasis and psoriatic arthritis. Biochem Pharmacol, 83 (12): 1583-90. [PMID:22257911] |
|
4. Schafer PH, Day RM. (2013)
Novel systemic drugs for psoriasis: mechanism of action for apremilast, a specific inhibitor of PDE4. J Am Acad Dermatol, 68 (6): 1041-2. [PMID:23680197] |
|
5. Schafer PH, Parton A, Capone L, Cedzik D, Brady H, Evans JF, Man HW, Muller GW, Stirling DI, Chopra R. (2014)
Apremilast is a selective PDE4 inhibitor with regulatory effects on innate immunity. Cell Signal, 26 (9): 2016-29. [PMID:24882690] |