GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: peptide boronate | PS-341 | Velcade®
bortezomib is an approved drug (FDA (2003), EMA (2004))
Compound class:
Synthetic organic
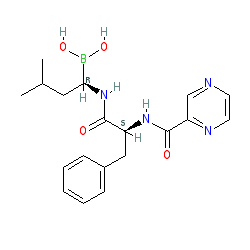
Comment: Bortezomib is a dipeptide (Phe-Leu), with a pyrazinoic acid protecting the N-terminus and a boronic acid replacing the C-terminal carboxylic acid. The boron atom is believed to interact with and inactivate the catalytic site on β subunits which form the active core of the proteasome, preferentially binding β5 active site [4]. Bortezomib is the first-in-class proteasome inhibitor to be approved for clinical use.
Proteasome activity is reviewed in [3]. The compound also has antimalarial activity. The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Alexander T, Sarfert R, Klotsche J, Kühl AA, Rubbert-Roth A, Lorenz HM, Rech J, Hoyer BF, Cheng Q, Waka A et al.. (2015)
The proteasome inhibitior bortezomib depletes plasma cells and ameliorates clinical manifestations of refractory systemic lupus erythematosus. Ann Rheum Dis, 74 (7): 1474-8. [PMID:25710470] |
|
2. Bonvini P, Zorzi E, Basso G, Rosolen A. (2007)
Bortezomib-mediated 26S proteasome inhibition causes cell-cycle arrest and induces apoptosis in CD-30+ anaplastic large cell lymphoma. Leukemia, 21 (4): 838-42. [PMID:17268529] |
|
3. Ciechanover A. (2005)
Intracellular protein degradation: from a vague idea, through the lysosome and the ubiquitin-proteasome system, and onto human diseases and drug targeting (Nobel lecture). Angew Chem Int Ed Engl, 44 (37): 5944-67. [PMID:16142822] |
|
4. Groll M, Berkers CR, Ploegh HL, Ovaa H. (2006)
Crystal structure of the boronic acid-based proteasome inhibitor bortezomib in complex with the yeast 20S proteasome. Structure, 14 (3): 451-6. [PMID:16531229] |
|
5. Jakez-Ocampo J, Atisha-Fregoso Y, Llorente L. (2015)
Refractory primary Sjögren syndrome successfully treated with bortezomib. J Clin Rheumatol, 21 (1): 31-2. [PMID:25539431] |
|
6. Mazepa MA, Raval JS, Moll S, Ma A, Park YA. (2014)
Bortezomib induces clinical remission and reduction of ADAMTS13 inhibitory antibodies in relapsed refractory idiopathic thrombotic thrombocytopenic purpura. Br J Haematol, 164 (6): 900-2. [PMID:24345005] |
|
7. Nakamura H, Watanabe M, Ban HS, Nabeyama W, Asai A. (2009)
Synthesis and biological evaluation of boron peptide analogues of Belactosin C as proteasome inhibitors. Bioorg Med Chem Lett, 19 (12): 3220-4. [PMID:19428245] |
|
8. Reynolds JM, El Bissati K, Brandenburg J, Günzl A, Mamoun CB. (2007)
Antimalarial activity of the anticancer and proteasome inhibitor bortezomib and its analog ZL3B. BMC Clin Pharmacol, 7: 13. [PMID:17956613] |
|
9. Wang Y, Zhou W, Zhang Z. (2015)
Successful treatment of warm-type haemolytic anaemia with bortezomib in a rituximab-failed systemic lupus erythematosus patient. Rheumatology (Oxford), 54 (1): 194-5. [PMID:25288787] |
|
10. Xie SC, Gillett DL, Spillman NJ, Tsu C, Luth MR, Ottilie S, Duffy S, Gould AE, Hales P, Seager BA et al.. (2018)
Target Validation and Identification of Novel Boronate Inhibitors of the Plasmodium falciparum Proteasome. J Med Chem, 61 (22): 10053-10066. [PMID:30373366] |
|
11. Zhang H, Liu Z, Huang L, Hou J, Zhou M, Huang X, Hu W, Liu Z. (2017)
The short-term efficacy of bortezomib combined with glucocorticoids for the treatment of refractory lupus nephritis. Lupus, 26 (9): 952-958. [PMID:28059023] |