|
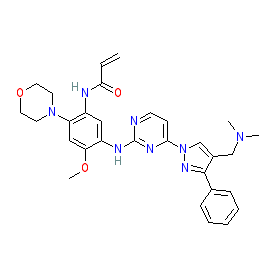
Synonyms: Compound 73 [WO2016060443A2] | GNS-1480 | GNS1480 | Leclaza® | YH-25448 | YH25448
lazertinib is an approved drug (S. Korea (2021))
Compound class:
Synthetic organic
Comment: Lazertinib (YH-25448, GNS-1480) is an oral, third-generation inhibitor of the aberrant activity of mutant EGFR kinases (EGFR T790M and the activating EGFR mutations Ex19del and L858R), that was originally developed by Yuhan Corporation for antineoplastic potential in cancers with (acquired) resistance to existing EGFR tyrosine kinase inhibitors [1], in particular for EGFR mutation positive, advanced non-small cell lung cancer (NSCLC). It is claimed in Yuhan's patent WO2016060443A2 as compound 73 [1].
In addition to predicted efficacy for the treatment of primary lung and extracranial lesions, lazertinib is expected to be beneficial for NSCLC brain metastases as it is able to cross the blood brain barrier. Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Suh B-C, Salgaonkar PD, Lee J, Koh JS, Song H-J, Lee IY, Lee J, Jung DS, Kim J-H, Kim S-W. (2016)
Compounds and compositions for modulating EGFR mutant kinase activities. Patent number: WO2016060443A2. Assignee: Yuhan Corporation. Priority date: 13/10/2014. Publication date: 21/04/2016. |