GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
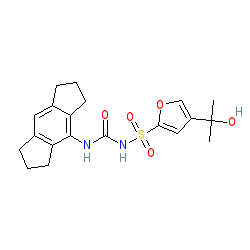
Synonyms: CP-456,773 | CP-456773 | CRID3 | MCC-950
Compound class:
Synthetic organic
Comment: MCC950 has been reported as a potent, selective and direct inhibitor of the NLRP3 inflammasome [4-5]. The compound binds directly at a motif within the NLRP3 protein's NACHT domain [2] and blocks its activation and inflammasome formation which ultimately reduces cleavage of IL-1β and IL-18 from their latent forms. It offers a useful tool for examination of NLRP3 biology and its potential for therapeutic intervention. MCC950 has demonstrated anti-inflammatory actions in numerous in vivo models of human diseases [3-4] and has been proposed as a therapeutic option for the treatment of NLRP3-associated diseases, including IL-1β/IL-18-driven inflammatory/autoinflammatory conditions [10,13] and other diseases that have an inflammatory component such as atherosclerosis [7,14], type 2 diabetes, Alzheimer's and Parkinson's diseases [8,12], cancer and pathologic inflammation that is a result of viral infection [1,11].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| MCC950 blocks canonical and non-canonical NLRP3-induced events, but spares NLRC4 and AIM2 inflammasome activity and TLR signaling. The potential of inflammasome inhibition as an anti-inflammatory modality is discussed in Fenini et al. (2017) [6]. |