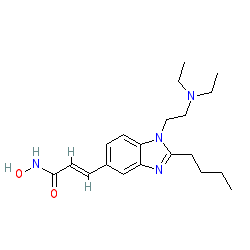
|
Synonyms: SB 939 | SB-939
Compound class:
Synthetic organic
Comment: Pracinostat is an orally available, potent pan-HDAC inhibitor [3].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| In March 2014, the US FDA granted pracinostat orphan designation for the treatment of the rare disease acute myelocytic leukemia (AML). In addition, the drug is currently being tested in Phase 2 trials in combination with azacitidine for the treatment of newly diagnosed AML in elderly patients (NCT01912274) and a further Phase 2 trial is assessing pracinostat + the Janus kinase inhibitor ruxolitinib in patients with myelofibrosis (MF) (see NCT02267278). |
Mechanism Of Action and Pharmacodynamic Effects  |
| HDAC inhibitors have shown clinical efficacy in AML, myelodysplastic syndrome and myeloproliferative neoplasms [2,4]. The HDAC inhibitor vorinostat was the first compound designed to inhibit HDACs to be approved for clinical use. HDAC inhibition increases acetylation of histone H3 and leads to downstream activation of the PI3K/Akt and MAPK/Ras signaling pathways and ultimately results in cell cycle arrest and apoptosis [1]. Mithraprabhu et al (2010) review the use of HDAC inhibitors in myeloproliferative neoplasms, and discuss the benefits of combination therapies with kinase inhibitors [2]. |