|
Synonyms: SB-207499
Compound class:
Synthetic organic
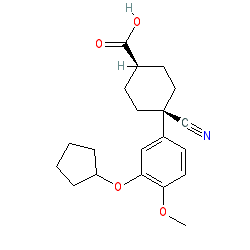
Comment: There is some ambiguity in the literature and on other database as to the exact structure of Cilomilast. The structure shown here matches that of the INN document, but the compound is represented on PubChem without specified stereochemistry by CID 151170.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Cilomilast is being investigated for the treatment of conditions such as chronic obstructive pulmonary disease (COPD), bronchopulmonary dysplasia and asthma. ClinicalTrials.gov lists one completed trial for this compound (NCT00103922) |
Mechanism Of Action and Pharmacodynamic Effects  |
| Cilomilast is a selective phosphodiesterase-4 (PDE4) inhibitor IC50 120nM [2]. This action is believed to confer the compound's anti-inflammatory activity. In vitro studies have shown that cilomilast suppresses LPS-induced TNF-α formation in isolated human monocytes (pIC50 7.0) and produces a modest inhibition of fMLP-induced degranulation of human neutrophils (pIC50 7.1) [1]. |