|
Synonyms: ATI-450 | ATI450 | CDD-450 | CDD450
Compound class:
Synthetic organic
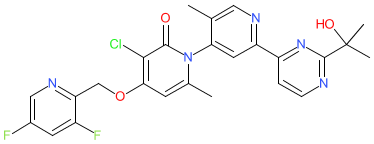
Comment: We obtained the chemical structure for zunsemetinib from WHO Proposed list 125 of July 2021. This mapped to PubChem CID 86291496. The compound is claimed in Confluence Life Sciences' patent US9115089B2 as kinase inhibitor with potential to treat p38 kinase-mediated diseases [2]. Confluence Life Sciences is a subsidiary of Aclaris Therapeutics. We speculate that zunsemetinib is the MAPK-activated protein kinase 2 (MK2) inhibitor ATI-450 (formerly CDD-450 [3]), that is included on Aclaris' pipeline page. Phase 1 safety and tolerability of ATI-450 were reported in June 2021 [1]. This article described ATI-450 as an inhibitor of p38α/MK2 pathway-mediated inflammatory signalling, but discloses only a partial chemical structure in a supplementary figure. The same incomplete chemical structure appears in [3]. ATI-450 binds with high affinity to the interface of the p38MAPK-MK2 complex and selectively inhibits p38MAPK-catalysed phosphorylation of MK2 which stabilises the inactive conformation of MK2, and subsequently reduces inflammatory cytokine levels [3].
|
|
|||||||||||||||||||||||||||||||||||
| No information available. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT04481685 | A Trial of Aclaris Therapeutics, Inc. (ATI)-450 in Patients With Moderate-severe Novel Coronavirus Disease 2019 (COVID-19) | Phase 2 Interventional | University of Kansas Medical Center | ||
| NCT04247815 | Study of ATI-450 Plus Methotrexate (MTX) vs MTX Alone in Patients With Moderate to Severe RA | Phase 2 Interventional | Aclaris Therapeutics, Inc. | ||
| NCT04524858 | Study of ATI-450 in Patients With Cryopyrin-Associated Periodic Syndrome (CAPS) | Phase 2 Interventional | Aclaris Therapeutics, Inc. | ||