GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Compound class:
Synthetic organic
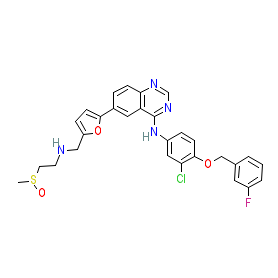
Comment: Selatinib (L-2) is a dual EGFR/HER2 receptor tyrosine kinase inhibitor that was developed for anti-cancer potential [1]. It is an anlogue of lapatinib and shares the same mechanism of action, but has an improved pharmacokinetic profile, with bioavailability at 187% of that of lapatinib. The tosylate salt form of selatinib is highly soluble. Selatinib is a 'pseudo' INN, that takes the form of an INN, but has not been submitted to the World Health Organisation for ratification.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| Bioactivity Comments |
| Selatinib inhibits proliferation of NCI-N87 gastric cancer-derived cells and BT-474 human breast tumour cells with IC50s of 29 and 22 nM respectively [1]. |
| Selectivity at catalytic receptors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||