GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
Contents:
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 863 | 8q24.12 | ENPP2 | ectonucleotide pyrophosphatase/phosphodiesterase 2 | |
| Mouse | - | 862 | 15 D1 | Enpp2 | ectonucleotide pyrophosphatase/phosphodiesterase 2 | |
| Rat | - | 887 | 7q32 | Enpp2 | ectonucleotide pyrophosphatase/phosphodiesterase 2 | |
| Gene and Protein Information Comments | ||||||
| The table provides information for human variant 1, which is the longest transcript and encodes the longest isoform (1, also known as alpha). Similarly for the mouse gene, which has 4 reported transcript variants, we provide information for the longest protein isoform. In humans and mice variant 2, which is translated to isoform β, is the major isoform found in tissue expression studies [7]. |
||||||
Previous and Unofficial Names  |
| ATX | PDNP2 | PD-IALPHA | lysoPLD |
Database Links  |
|
| Alphafold | Q13822 (Hs), Q9R1E6 (Mm), Q64610 (Rn) |
| BRENDA | 3.1.4.39 |
| CATH/Gene3D | 3.40.720.10 |
| ChEMBL Target | CHEMBL3691 (Hs), CHEMBL3826871 (Mm), CHEMBL3826870 (Rn) |
| Ensembl Gene | ENSG00000136960 (Hs), ENSMUSG00000022425 (Mm), ENSRNOG00000004089 (Rn) |
| Entrez Gene | 5168 (Hs), 18606 (Mm), 84050 (Rn) |
| Human Protein Atlas | ENSG00000136960 (Hs) |
| KEGG Enzyme | 3.1.4.39 |
| KEGG Gene | hsa:5168 (Hs), mmu:18606 (Mm), rno:84050 (Rn) |
| OMIM | 601060 (Hs) |
| Pharos | Q13822 (Hs) |
| RefSeq Nucleotide | NM_006209 (Hs), NM_001040092 (Hs), NM_001130863 (Hs), NM_001136077 (Mm), NM_015744 (Mm), NM_001285995 (Mm), NM_001285994 (Mm), NM_057104 (Rn) |
| RefSeq Protein | NP_001035181 (Hs), NP_001124335 (Hs), NP_006200 (Hs), NP_001272923 (Mm), NP_001272924 (Mm), NP_056559 (Mm), NP_001129549 (Mm), NP_476445 (Rn) |
| UniProtKB | Q13822 (Hs), Q9R1E6 (Mm), Q64610 (Rn) |
| Wikipedia | ENPP2 (Hs) |
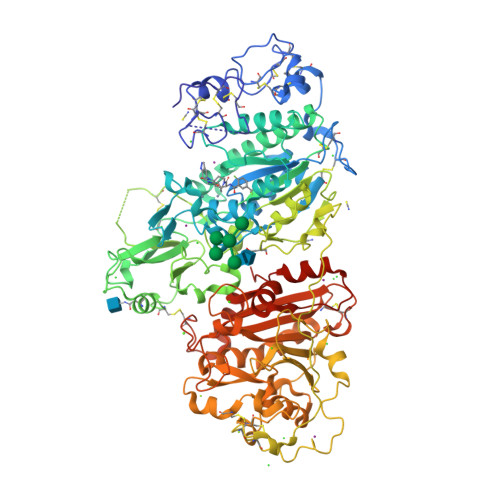
Selected 3D Structures  |
|||||||||||||
|
|
||||||||||||
Enzyme Reaction  |
||||||||||
|
||||||||||
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| View species-specific inhibitor tables | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inhibitor Comments | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ATX inhibitors are being investigated for potential anti-fibrotic activity. ATX inhibitor development and exemplar compounds are discussed in [2]. Ziritaxestat (GLPG1690) has completed a first-in-human Phase 1 clinical trial (NCT02179502). Phase 2 trial NCT02738801 is evaluating the safety, tolerability, PK and PD of GLPG1690 in patients with idiopathic pulmonary fibrosis (IPF). However, example 31 (24) (a.k.a. Ex_31) failed to show any anti-fibrotic effects in two models of advanced liver fibrosis in rats [1]. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| Autotaxin is up-regulated in many inflammatory conditions, including cancer, arthritis, fibrotic diseases and multiple sclerosis. The product of autotaxin activity, LPA, has proliferative, chemotactic and angiogenic actions. Inhibitors of the ATX-LPA axis are being investigated as novel pharmaceuticals [2]. |
Tissue Distribution 
|
||||||||
|
Physiological Functions 
|
||||||||
|
||||||||
|
References
1. Baader M, Bretschneider T, Broermann A, Rippmann JF, Stierstorfer B, Kuttruff CA, Mark M. (2018) Characterization of the properties of a selective, orally bioavailable autotaxin inhibitor in preclinical models of advanced stages of liver fibrosis. Br J Pharmacol, 175 (4): 693-707. [PMID:29197066]
2. Castagna D, Budd DC, Macdonald SJ, Jamieson C, Watson AJ. (2016) Development of Autotaxin Inhibitors: An Overview of the Patent and Primary Literature. J Med Chem, 59 (12): 5604-21. [PMID:26745766]
3. Clair T, Aoki J, Koh E, Bandle RW, Nam SW, Ptaszynska MM, Mills GB, Schiffmann E, Liotta LA, Stracke ML. (2003) Autotaxin hydrolyzes sphingosylphosphorylcholine to produce the regulator of migration, sphingosine-1-phosphate. Cancer Res, 63 (17): 5446-53. [PMID:14500380]
4. Cuozzo JW, Clark MA, Keefe AD, Kohlmann A, Mulvihill M, Ni H, Renzetti LM, Resnicow DI, Ruebsam F, Sigel EA et al.. (2020) Novel Autotaxin Inhibitor for the Treatment of Idiopathic Pulmonary Fibrosis: A Clinical Candidate Discovered Using DNA-Encoded Chemistry. J Med Chem, 63 (14): 7840-7856. [PMID:32584034]
5. Desroy N, Housseman C, Bock X, Joncour A, Bienvenu N, Cherel L, Labeguere V, Rondet E, Peixoto C, Grassot JM et al.. (2017) Discovery of 2-[[2-Ethyl-6-[4-[2-(3-hydroxyazetidin-1-yl)-2-oxoethyl]piperazin-1-yl]-8-methylimidazo[1,2-a]pyridin-3-yl]methylamino]-4-(4-fluorophenyl)thiazole-5-carbonitrile (GLPG1690), a First-in-Class Autotaxin Inhibitor Undergoing Clinical Evaluation for the Treatment of Idiopathic Pulmonary Fibrosis. J Med Chem, 60 (9): 3580-3590. [PMID:28414242]
6. Gierse J, Thorarensen A, Beltey K, Bradshaw-Pierce E, Cortes-Burgos L, Hall T, Johnston A, Murphy M, Nemirovskiy O, Ogawa S et al.. (2010) A novel autotaxin inhibitor reduces lysophosphatidic acid levels in plasma and the site of inflammation. J Pharmacol Exp Ther, 334 (1): 310-7. [PMID:20392816]
7. Giganti A, Rodriguez M, Fould B, Moulharat N, Cogé F, Chomarat P, Galizzi JP, Valet P, Saulnier-Blache JS, Boutin JA et al.. (2008) Murine and human autotaxin alpha, beta, and gamma isoforms: gene organization, tissue distribution, and biochemical characterization. J Biol Chem, 283 (12): 7776-89. [PMID:18175805]
8. Guckian K, Kumaravel G, Ma B, Mi S, Peng H, Shao Z, Sun L, Taveras A, Wang D, Xin Z, Zhang L. (2014) Atx modulating agents. Patent number: WO2014018881A1. Assignee: Biogen Idec Ma Inc.. Priority date: 27/07/2012. Publication date: 30/01/2014.
9. Hert J, Hunziker D, Mattei P, Mauser H, Tang G, Wang L. (2014) New octahydro-pyrrolo[3,4-c]-pyrrole derivatives and analogs thereof as autotaxin inhibitors. Patent number: WO2014139978 A1. Assignee: Hoffmann-La Roche Inc.. Priority date: 12/03/2013. Publication date: 18/09/2014.
10. Kuttruff CA, Ferrara M, Bretschneider T, Hoerer S, Handschuh S, Nosse B, Romig H, Nicklin P, Roth GJ. (2017) Discovery of BI-2545: A Novel Autotaxin Inhibitor That Significantly Reduces LPA Levels in Vivo. ACS Med Chem Lett, 8 (12): 1252-1257. [PMID:29259743]
11. Long S, Thorarensen A, Schnute ME. (2013) Pyrimidine and pyridine derivatives useful in therapy. Patent number: WO2013054185 A1. Assignee: Pfizer, Inc.. Priority date: 13/10/2011. Publication date: 18/04/2013.
12. Nikolaou A, Ninou I, Kokotou MG, Kaffe E, Afantitis A, Aidinis V, Kokotos G. (2018) Hydroxamic Acids Constitute a Novel Class of Autotaxin Inhibitors that Exhibit in Vivo Efficacy in a Pulmonary Fibrosis Model. J Med Chem, 61 (8): 3697-3711. [PMID:29620892]
13. Shah P, Cheasty A, Foxton C, Raynham T, Farooq M, Gutierrez IF, Lejeune A, Pritchard M, Turnbull A, Pang L et al.. (2016) Discovery of potent inhibitors of the lysophospholipase autotaxin. Bioorg Med Chem Lett, 26 (22): 5403-5410. [PMID:27780639]
14. St-Cœur PD, Ferguson D, Morin Jr P, Touaibia M. (2013) PF-8380 and closely related analogs: synthesis and structure-activity relationship towards autotaxin inhibition and glioma cell viability. Arch Pharm (Weinheim), 346 (2): 91-7. [PMID:23300119]
How to cite this page
LPA synthesis: autotaxin. Last modified on 19/08/2024. Accessed on 02/05/2026. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetoimmunopharmacology.org/GRAC/ObjectDisplayForward?objectId=2901.

 Target has curated data in GtoImmuPdb
Target has curated data in GtoImmuPdb