GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
Contents:
- Gene and Protein Information
- Previous and Unofficial Names
- Database Links
- Selected 3D Structures
- Enzyme Reaction
- Inhibitors
- Antibodies
- Immunopharmacology Comments
- Immuno Process Associations
- Immuno Disease Associations
- Clinically-Relevant Mutations and Pathophysiology
- References
- How to cite this page
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 707 | 20q13.12 | MMP9 | matrix metallopeptidase 9 | |
| Mouse | - | 730 | 2 85.27 cM | Mmp9 | matrix metallopeptidase 9 | |
| Rat | - | 708 | 3q42 | Mmp9 | matrix metallopeptidase 9 | |
Previous and Unofficial Names  |
| GELB | gelatinase B | B/MMP9 | CLG4B | matrix metallopeptidase 9 (gelatinase B, 92kDa gelatinase, 92kDa type IV collagenase) |
Database Links  |
|
| Specialist databases | |
| MEROPS | M10.004 (Hs) |
| Other databases | |
| Alphafold | P14780 (Hs), P41245 (Mm), P50282 (Rn) |
| BRENDA | 3.4.24.35 |
| CATH/Gene3D | 2.10.10.10, 2.110.10.10, 3.40.390.10, 1.10.101.10 |
| ChEMBL Target | CHEMBL321 (Hs), CHEMBL2214 (Mm), CHEMBL3870 (Rn) |
| DrugBank Target | P14780 (Hs) |
| Ensembl Gene | ENSG00000100985 (Hs), ENSMUSG00000017737 (Mm), ENSRNOG00000017539 (Rn) |
| Entrez Gene | 4318 (Hs), 17395 (Mm), 81687 (Rn) |
| Human Protein Atlas | ENSG00000100985 (Hs) |
| KEGG Enzyme | 3.4.24.35 |
| KEGG Gene | hsa:4318 (Hs), mmu:17395 (Mm), rno:81687 (Rn) |
| OMIM | 120361 (Hs) |
| Orphanet | ORPHA208342 (Hs) |
| Pharos | P14780 (Hs) |
| RefSeq Nucleotide | NM_004994 (Hs), NM_013599 (Mm), NM_031055 (Rn) |
| RefSeq Protein | NP_004985 (Hs), NP_038627 (Mm), NP_112317 (Rn) |
| UniProtKB | P14780 (Hs), P41245 (Mm), P50282 (Rn) |
| Wikipedia | MMP9 (Hs) |
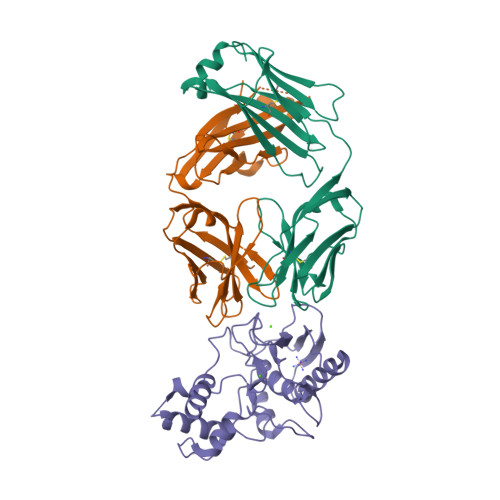
Selected 3D Structures  |
|||||||||||||
|
|
||||||||||||
Enzyme Reaction  |
||||
|
||||
Download all structure-activity data for this target as a CSV file 
| Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | View all chemical structures | Click column headers to sort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Inhibitor Comments | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SL422 is a broad-spectrum inhibitor of MMP enzymes [18]. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antibodies | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | ||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| Mucosal up-regulation of MMP-9 correlates with the severity of inflammation in ulcerative colitis, and may be responsible for the mucosal damage in active ulcerative colitis [9]. |
| Immuno Process Associations | |||||||||
|
|||||||||
|
| Immuno Disease Associations | ||||||||||
|
||||||||||
|
||||||||||
|
Clinically-Relevant Mutations and Pathophysiology 
|
||||||||||||
|
||||||||||||
|
||||||||||||
References
1. Anthony DC, Ferguson B, Matyzak MK, Miller KM, Esiri MM, Perry VH. (1997) Differential matrix metalloproteinase expression in cases of multiple sclerosis and stroke. Neuropathol Appl Neurobiol, 23 (5): 406-15. [PMID:9364466]
2. Appleby TC, Greenstein AE, Hung M, Liclican A, Velasquez M, Villaseñor AG, Wang R, Wong MH, Liu X, Papalia GA et al.. (2017) Biochemical characterization and structure determination of a potent, selective antibody inhibitor of human MMP9. J Biol Chem, 292 (16): 6810-6820. [PMID:28235803]
3. AstraZeneca. AZD1236. Accessed on 31/10/2014. Modified on 31/10/2014. Open Innovation, http://openinnovation.astrazeneca.com/what-we-offer/compound/azd1236/
4. Benesová Y, Vasku A, Novotná H, Litzman J, Stourac P, Beránek M, Kadanka Z, Bednarík J. (2009) Matrix metalloproteinase-9 and matrix metalloproteinase-2 as biomarkers of various courses in multiple sclerosis. Mult Scler, 15 (3): 316-22. [PMID:19153173]
5. Brown S, Bernardo MM, Li Z-H, Kotra LP, Tanaka Y, Fridman R, Mobashery S. (2000) Potent and Selective Mechanism-Based Inhibition of Gelatinases. J Am Chem Soc, 122: 6799-6800.
6. Cataldo D, Munaut C, Noël A, Frankenne F, Bartsch P, Foidart JM, Louis R. (2000) MMP-2- and MMP-9-linked gelatinolytic activity in the sputum from patients with asthma and chronic obstructive pulmonary disease. Int Arch Allergy Immunol, 123 (3): 259-67. [PMID:11112863]
7. Jin Y, Roycik MD, Bosco DB, Cao Q, Constantino MH, Schwartz MA, Sang QX. (2013) Matrix metalloproteinase inhibitors based on the 3-mercaptopyrrolidine core. J Med Chem, 56 (11): 4357-73. [PMID:23631440]
8. Kirkegaard T, Hansen A, Bruun E, Brynskov J. (2004) Expression and localisation of matrix metalloproteinases and their natural inhibitors in fistulae of patients with Crohn's disease. Gut, 53 (5): 701-9. [PMID:15082589]
9. Lakatos G, Sipos F, Miheller P, Hritz I, Varga MZ, Juhász M, Molnár B, Tulassay Z, Herszényi L. (2012) The behavior of matrix metalloproteinase-9 in lymphocytic colitis, collagenous colitis and ulcerative colitis. Pathol Oncol Res, 18 (1): 85-91. [PMID:21678108]
10. Ma D, Wu W, Yang G, Li J, Li J, Ye Q. (2004) Tetrahydroisoquinoline based sulfonamide hydroxamates as potent matrix metalloproteinase inhibitors. Bioorg Med Chem Lett, 14 (1): 47-50. [PMID:14684295]
11. Marshall DC, Lyman SK, McCauley S, Kovalenko M, Spangler R, Liu C, Lee M, O'Sullivan C, Barry-Hamilton V, Ghermazien H et al.. (2015) Selective Allosteric Inhibition of MMP9 Is Efficacious in Preclinical Models of Ulcerative Colitis and Colorectal Cancer. PLoS ONE, 10 (5): e0127063. [PMID:25961845]
12. Moriyama H, Tsukida T, Inoue Y, Yokota K, Yoshino K, Kondo H, Miura N, Nishimura S. (2004) Azasugar-based MMP/ADAM inhibitors as antipsoriatic agents. J Med Chem, 47 (8): 1930-8. [PMID:15055993]
13. MRC. AZD6605 Matrix metallopeptidase 13 (MMP13) inhibitor. Accessed on 28/10/2014. Modified on 28/10/2014. MRC/AstraZeneca: Mechanisms of Disease Call, http://webarchive.nationalarchives.gov.uk/20120104105854/http://www.mrc.ac.uk/consumption/groups/public/documents/content/mrc008371.pdf
14. Nguyen TT, Ding D, Wolter WR, Pérez RL, Champion MM, Mahasenan KV, Hesek D, Lee M, Schroeder VA, Jones JI et al.. (2018) Validation of Matrix Metalloproteinase-9 (MMP-9) as a Novel Target for Treatment of Diabetic Foot Ulcers in Humans and Discovery of a Potent and Selective Small-Molecule MMP-9 Inhibitor That Accelerates Healing. J Med Chem, 61 (19): 8825-8837. [PMID:30212201]
15. Nuti E, Casalini F, Santamaria S, Gabelloni P, Bendinelli S, Da Pozzo E, Costa B, Marinelli L, La Pietra V, Novellino E et al.. (2011) Synthesis and biological evaluation in U87MG glioma cells of (ethynylthiophene)sulfonamido-based hydroxamates as matrix metalloproteinase inhibitors. Eur J Med Chem, 46 (7): 2617-29. [PMID:21514700]
16. Rasmussen HS, McCann PP. (1997) Matrix metalloproteinase inhibition as a novel anticancer strategy: a review with special focus on batimastat and marimastat. Pharmacol Ther, 75 (1): 69-75. [PMID:9364582]
17. Takeuchi T, Hayashi M, Tamita T, Nomura Y, Kojima N, Mitani A, Takeda T, Hitaka K, Kato Y, Kamitani M et al.. (2022) Discovery of Aryloxyphenyl-Heptapeptide Hybrids as Potent and Selective Matrix Metalloproteinase-2 Inhibitors for the Treatment of Idiopathic Pulmonary Fibrosis. J Med Chem, 65 (12): 8493-8510. [PMID:35687819]
18. Xue CB, Voss ME, Nelson DJ, Duan JJ, Cherney RJ, Jacobson IC, He X, Roderick J, Chen L, Corbett RL et al.. (2001) Design, synthesis, and structure-activity relationships of macrocyclic hydroxamic acids that inhibit tumor necrosis factor alpha release in vitro and in vivo. J Med Chem, 44 (16): 2636-60. [PMID:11472217]
19. Yong VW, Zabad RK, Agrawal S, Goncalves Dasilva A, Metz LM. (2007) Elevation of matrix metalloproteinases (MMPs) in multiple sclerosis and impact of immunomodulators. J Neurol Sci, 259: 79-84. [PMID:17382965]
How to cite this page
M10: Matrix metallopeptidase: MMP9. Last modified on 12/07/2022. Accessed on 23/05/2026. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetoimmunopharmacology.org/GRAC/ObjectDisplayForward?objectId=1633.

 Target has curated data in GtoImmuPdb
Target has curated data in GtoImmuPdb