|
Synonyms: example 352 [WO2007058338] | MM36 | Moizerto® | OPA-15406
difamilast is an approved drug (Japan (2021))
Compound class:
Synthetic organic
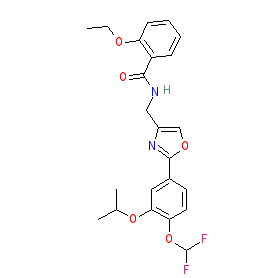
Comment: Difamilast (OPA-15406) is a topical, selective, nonsteroidal PDE4 inhibitor that is being developed for the treatment of atopic dermatitis [1-3]. The chemical structure is claimed as example 352 in patent WO2007058338 [4].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Freitas E, Torres T. (2023)
New molecules for atopic dermatitis treatment beyond biological therapy. Curr Opin Allergy Clin Immunol, 23 (3): 210-215. [PMID:37185825] |
|
2. Hanifin JM, Ellis CN, Frieden IJ, Fölster-Holst R, Stein Gold LF, Secci A, Smith AJ, Zhao C, Kornyeyeva E, Eichenfield LF. (2016)
OPA-15406, a novel, topical, nonsteroidal, selective phosphodiesterase-4 (PDE4) inhibitor, in the treatment of adult and adolescent patients with mild to moderate atopic dermatitis (AD): A phase-II randomized, double-blind, placebo-controlled study. J Am Acad Dermatol, 75 (2): 297-305. [PMID:27189825] |
|
3. Hiyama H, Arichika N, Okada M, Koyama N, Tahara T, Haruta J. (2023)
Pharmacological Profile of Difamilast, a Novel Selective Phosphodiesterase 4 Inhibitor, for Topical Treatment of Atopic Dermatitis. J Pharmacol Exp Ther, 386 (1): 45-55. [PMID:37041087] |
|
4. Okada M, Kato M, Sato N, Uno T, Kitagaki H, Haruta J, Hiyama H, Shibata T. (2007)
Oxazole compound and pharmaceutical composition. Patent number: WO2007058338. Assignee: Otsuka Pharmaceutical Co., Ltd.. Priority date: 24/05/2007. Publication date: 15/11/2005. |
|
5. Saeki H, Imamura T, Yokota D, Tsubouchi H. (2022)
Difamilast Ointment in Japanese Adult and Pediatric Patients with Atopic Dermatitis: A Phase III, Long-Term, Open-Label Study. Dermatol Ther (Heidelb), 12 (7): 1589-1601. [PMID:35716332] |