|
Compound class:
Natural product
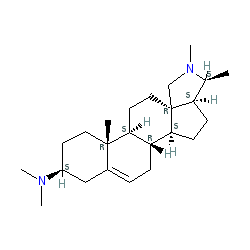
Comment: Conessine is an alkaloid obtained from the bark and seeds of Holarrhena antidysenterica trees. It binds with high affinity to rat and human histamine H3 receptors in radio-ligand binding assays, acting as a receptor antagonist [2-3]. H3 antagonists are being researched as potential drugs for the treatment of neurodegenerative conditions and for their stimulant and nootropic effects. For example, pitolisant is in clinical trial for Parkinson's disease, narcolepsy, cataplexy and excessive daytime sleepiness.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Panula P, Chazot PL, Cowart M, Gutzmer R, Leurs R, Liu WL, Stark H, Thurmond RL, Haas HL. (2015)
International Union of Basic and Clinical Pharmacology. XCVIII. Histamine Receptors. Pharmacol Rev, 67 (3): 601-55. [PMID:26084539] |
|
2. Santora VJ, Covel JA, Hayashi R, Hofilena BJ, Ibarra JB, Pulley MD, Weinhouse MI, Sengupta D, Duffield JJ, Semple G et al.. (2008)
A new family of H3 receptor antagonists based on the natural product Conessine. Bioorg Med Chem Lett, 18 (4): 1490-4. [PMID:18194865] |
|
3. Zhao C, Sun M, Bennani YL, Gopalakrishnan SM, Witte DG, Miller TR, Krueger KM, Browman KE, Thiffault C, Wetter J et al.. (2008)
The alkaloid conessine and analogues as potent histamine H3 receptor antagonists. J Med Chem, 51 (17): 5423-30. [PMID:18683917] |