|
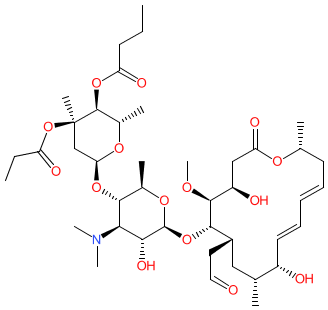
Synonyms: Ricamycin® | TMS-19-Q
rokitamycin is an approved drug
Compound class:
Synthetic organic
Comment: Rokitamycin is a 16-membered semisynthetic macrolide antibacterial compound [3].
|
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Braga PC. (2002)
Rokitamycin: bacterial resistance to a 16-membered ring macrolide differs from that to 14- and 15-membered ring macrolides. J Chemother, 14 (2): 115-31. [PMID:12017366] |
|
2. Cinco M, Padovan D, Stinco G, Trevisan G. (1995)
In vitro activity of rokitamycin, a new macrolide, against Borrelia burgdorferi. Antimicrob Agents Chemother, 39 (5): 1185-6. [PMID:7625812] |
|
3. Sakakibara H, Okekawa O, Fujiwara T, Otani M, Omura S. (1981)
Acyl derivatives of 16-membered macrolides. I. Synthesis and biological properties of 3"-O-propionylleucomycin A5 (TMS-19-Q). J Antibiot (Tokyo), 34 (8): 1001-10. [PMID:7319915] |