|
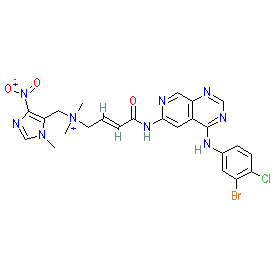
Synonyms: PR610 | tarloxotinib cation | TH-4000
Compound class:
Synthetic organic
Comment: Tarloxotinib (TH-4000) is a hypoxia-activated prodrug (HAP) [1] being developed by Threshold Pharmaceuticals. Delivered as the bromide salt (PubChem CID 51038315) as described in its INN record. The compound undergoes activation selectively within the hypoxic tumour compartment to produce an irreversible pan-ErbB tyrosine-kinase inhibitor, represented by TH-4000E [1]. The 4-nitroimidazole trigger on the tertiary amine side chain facilitates activation. This is one of the first molecularly targeted HAPs to be developed.
|
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Tarloxotinib (TH-4000) failed to show clinical efficacy in Phase 2 clinical trial for EGFR mutant, T790M-negative NSCLC (NCT02454842) and recurrent or metastatic squamous cell carcinoma of the head and neck or skin (NCT02449681). Development was subsequently terminated. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT02449681 | Study for Treatment of Patients With Recurrent or Metastatic SCCHN or SCCS | Phase 2 Interventional | Threshold Pharmaceuticals | ||
| NCT02454842 | Study for Treatment of Patients With EGFR Mutant, T790M-negative NSCLC | Phase 2 Interventional | Threshold Pharmaceuticals | ||