|
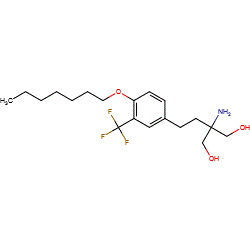
Synonyms: Example 1 [US20090137530] | MT-1303 | MT1303
Compound class:
Synthetic organic
Comment: Amiselimod (MT-1303) is claimed in patent US20090137530 (Compound 1-3 is the hydrochloride used experimentally) as a potential immunomodulatory agent [1]. It is a sphingosine-1-phosphate (SIP) receptor prodrug agonist, similar in action to the approved MS drug fingolimod, but with improved receptor subtype selectivity [3].
Selective SIP1 receptor agonists are being investigated for immunomodulatory/immunosuppresant action in MS and other autoimmune conditions [2]. |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Amiselimod (MT-1303) has completed Phase 2 dose-finding clinical trials in patients with relapsing-remitting multiple sclerosis (NCT01742052) and moderate to severe chronic plaque psoriasis (NCT01987843). Phase 2 trials in Crohn's disease patients are underway. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT01742052 | Dose-finding Study of MT-1303 | Phase 2 Interventional | Mitsubishi Tanabe Pharma Corporation | ||
| NCT01987843 | Dose-finding Study of MT-1303 in Subjects With Moderate to Severe Chronic Plaque Psoriasis | Phase 2 Interventional | Mitsubishi Tanabe Pharma Corporation | ||