GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
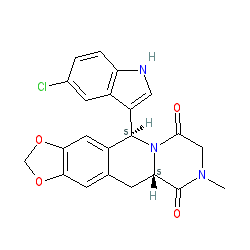
Compound class:
Synthetic organic
|
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Mechanism Of Action and Pharmacodynamic Effects  |
| PIP5K1A is responsible for the production of PIP2 (phosphatidylinositol 4,5-bisphosphate, PtdIns-4,5-P2) [2], which is subsequently converted to PIP3 (phosphatidylinositol 3,4,5-trisphosphate, PtdIns(3,4,5)P3), an activator of the AKT family of serine/threonine kinases [1,4]. Elevated AKT activity is frequently observed in human cancer cells. Inhibition of PIP5K1A would therefore be expected to reduce PIP2 levels and AKT activity, with potential anti-cancer effects. |