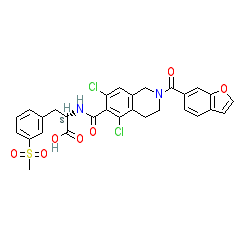
|
Synonyms: compound 1g [PMID: 24900456] | SAR 1118 | SAR-1118 | SAR-1118-023 | Xiidra®
lifitegrast is an approved drug (FDA (2016))
Compound class:
Synthetic organic
Comment: It is postulated that lifitegrast (SAR 1118) interacts with the lymphocyte function-associated antigen-1 (LFA-1) to inhbit the LFA-1/ICAM-1 adhesion interaction and was approved for the treatment of Dry Ey in 2016 [7,9]. LFA-1 is a heterodimeric member of the β2 integrin family which is essential for adhesion, migration, and proliferation of T cells at sites of inflammation, and is involved in T cell specific immune responses through the formation of adhesive interactions and immunological synapses. Intercellular adhesion molecule‑1 (ICAM‑1) is the cognate ligand of LFA-1.The compound formulation is claimed in patent WO2014100135 [7].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Lifitegrast is a topical integrin antagonist approved by the US FDA (July 2016) for the treatment of keratoconjunctivitis sicca (dry eye disease) [8-9]. Phase 3 trial results are published in [8]. |
Mechanism Of Action and Pharmacodynamic Effects  |
| Dry eye disease is believed to be a chronic inflammatory condition [5]. The disease is accompanied by an increase of ICAM-1 expression, which is postulated to underpin the LFA-1/ICAM-1 interaction which supports the pathological inflammatory and specific T-cell regulated immune responses in this condition. Although the mechanism of action of lifitegrast in dry eye disease is not fully resolved, the drug has been shown to antagonise the interaction between ICAM-1 and LFA-1 to bring about a reduction in the signs and symptoms of the condition [6]. Giblin and Lemieux (2006) [3] review LFA-1 as a key regulator of immune function and discuss the mechanisms employed by the different types of LFA-1-based therapeutics in development [1]. Descriptive content in patent WO2015189265 [2] alludes to lifitegrast being an α/β I allosteric LFA-1 inhibitor; this class of inhibitors are ligand mimetics based on amino acid sequence of the LFA-1 binding region of ICAM, which are found to compete for binding of an internal ligand of LFA-1 beta chain. A more extensive review of integrin modulators in the patent literature is provided by Kapp et al. (2013) [4]. |