|
Synonyms: RV-6153
Compound class:
Synthetic organic
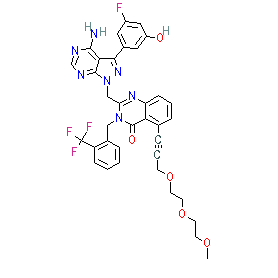
Comment: RV6153 is a clinical stage PI3Kγδ inhibitor [1] that is being developed by Janssen Biotech subsidiary RespiVert. The chemical structure is claimed in patent US9227977B2 [2]. It is a derivative of RV1729. RV6153 is selective for PI3Kγδ over the α and β enzyme isoforms.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| A Phase 1 study of RV6153 in healthy volunteers and asthma patients (NCT02517359) was terminated as the preset pharmacokinetic stopping criteria were met at lower doses than expected. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT02517359 | A Study to Investigate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of Single and Repeat Doses of RV6153 | Phase 1 Interventional | Respivert Ltd | ||