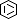GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
Contents:
Gene and Protein Information  |
||||||
| Species | TM | AA | Chromosomal Location | Gene Symbol | Gene Name | Reference |
| Human | - | 147 | 18q12.1 | TTR | transthyretin | |
| Mouse | - | 147 | 18 11.47 cM | Ttr | transthyretin | |
| Rat | - | 147 | 18p12 | Ttr | transthyretin | |
Previous and Unofficial Names  |
|
| carpal tunnel syndrome 1 [4] | CTS1 | PALB | prealbumin, amyloidosis type I | |
Database Links  |
|
| Alphafold | P02766 (Hs), P07309 (Mm), P02767 (Rn) |
| CATH/Gene3D | 2.60.40.180 |
| ChEMBL Target | CHEMBL3194 (Hs), CHEMBL5169111 (Mm), CHEMBL2151 (Rn) |
| Ensembl Gene | ENSG00000118271 (Hs), ENSMUSG00000061808 (Mm), ENSRNOG00000016275 (Rn) |
| Entrez Gene | 7276 (Hs), 22139 (Mm), 24856 (Rn) |
| Human Protein Atlas | ENSG00000118271 (Hs) |
| KEGG Gene | hsa:7276 (Hs), mmu:22139 (Mm), rno:24856 (Rn) |
| OMIM | 176300 (Hs) |
| Pharos | P02766 (Hs) |
| RefSeq Nucleotide | NM_000371 (Hs), NM_013697 (Mm), NM_012681 (Rn) |
| RefSeq Protein | NP_000362 (Hs), NP_038725 (Mm), NP_036813 (Rn) |
| SynPHARM | 82072 (in complex with tafamidis) |
| UniProtKB | P02766 (Hs), P07309 (Mm), P02767 (Rn) |
| Wikipedia | TTR (Hs) |
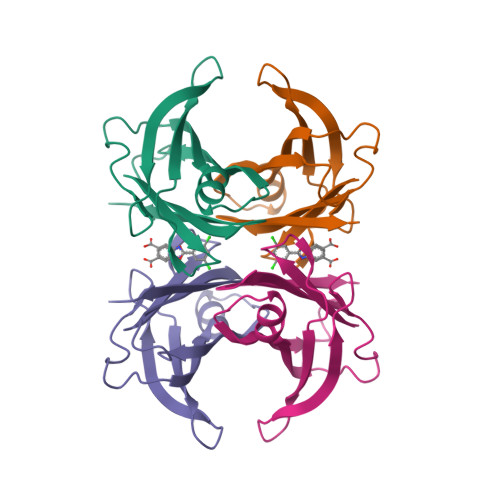
Selected 3D Structures  |
|||||||||||||
|
|
||||||||||||
|
|
||||||||||||

|
|
||||||||||||
| Other Binding Ligands | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Clinically-Relevant Mutations and Pathophysiology 
|
||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||
| Gene Expression and Pathophysiology Comments | |
| Excess production and accumulation of transthyretin (TTR) causes hereditary transthyretin-mediated amyloidosis. Drugs that directly inhibit TTR expression (of both wild type and mutant TTR) in the liver, are now approved for use in clinical practice. |
| General Comments |
| Three drugs that disrupt TTR expression have been approved for clinical use, as a mechanism to treat hereditary transthyretin-mediated amyloidosis: inotersen (Tegsedi®) is an antisense oligonucleotide, and patisiran (Onpattro®) and vutrisiran (Amvuttra®) are double-stranded small interfering RNAs. As an alternative gene-level intervention, Intellia Therapeutics have conducted a first-in-human trial of their in vivo CRISPR/Cas9 TTR genome editing candidate, NTLA-2001 (NCT04601051) [6]. NTLA-2001 reduced serum levels of transthyretin. The orally administered, small molecule TTR stabilising drugs tafamidis and acoramidis are authorised for the treament of transthyretin amyloid cardiomyopathy (ATTR-CM). TTR stabilisers inhibit dissociation of the tetrameric protein complex into the monomeric units that can misfold and accumulate as toxic TTR amyloid deposits within organs. |
References
1. Benson MD. (1991) Inherited amyloidosis. J Med Genet, 28 (2): 73-8. [PMID:1848299]
2. Bulawa CE, Connelly S, Devit M, Wang L, Weigel C, Fleming JA, Packman J, Powers ET, Wiseman RL, Foss TR et al.. (2012) Tafamidis, a potent and selective transthyretin kinetic stabilizer that inhibits the amyloid cascade. Proc Natl Acad Sci USA, 109 (24): 9629-34. [PMID:22645360]
3. Iakovleva I, Brännström K, Nilsson L, Gharibyan AL, Begum A, Anan I, Walfridsson M, Sauer-Eriksson AE, Olofsson A. (2015) Enthalpic Forces Correlate with the Selectivity of Transthyretin-Stabilizing Ligands in Human Plasma. J Med Chem, 58 (16): 6507-15. [PMID:26214366]
4. Murakami T, Tachibana S, Endo Y, Kawai R, Hara M, Tanase S, Ando M. (1994) Familial carpal tunnel syndrome due to amyloidogenic transthyretin His 114 variant. Neurology, 44 (2): 315-8. [PMID:8309582]
5. Penchala SC, Connelly S, Wang Y, Park MS, Zhao L, Baranczak A, Rappley I, Vogel H, Liedtke M, Witteles RM et al.. (2013) AG10 inhibits amyloidogenesis and cellular toxicity of the familial amyloid cardiomyopathy-associated V122I transthyretin. Proc Natl Acad Sci USA, 110 (24): 9992-7. [PMID:23716704]
6. Yadav JD, Othee H, Chan KA, Man DC, Belliveau PP, Towle J. (2021) Transthyretin Amyloid Cardiomyopathy-Current and Future Therapies. Ann Pharmacother, 55 (12): 1502-1514. [PMID:33685242]
How to cite this page
Transthyretin: transthyretin. Last modified on 27/02/2025. Accessed on 29/04/2026. IUPHAR/BPS Guide to PHARMACOLOGY, https://www.guidetoimmunopharmacology.org/GRAC/ObjectDisplayForward?objectId=2851.