GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
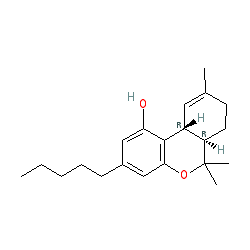
Abbreviated name: THC
Synonyms: Δ9-THC | Abbott 40566 | delta9-THC | Marinol® | tetrahydrocannabinol
Δ9-tetrahydrocannabinol is an approved drug (FDA (1985))
Compound class:
Natural product
Comment: Δ9-tetrahydrocannabinol is the principle active ingredient derived from the cannabis plant (Cannabis sativa). Dronabinol is the INN for the pure, synthetically produced (-)-trans-Δ9-tetrahydrocannabinol isomer of tetrahydrocannabinol (shown here).
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖
View more information in the IUPHAR Pharmacology Education Project: delta-9-tetrahydrocannabinol |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Used in the treatment of anorexia in patients with AIDS, and treatment of nausea and vomiting associated with cancer chemotherapy in patients who have not reposnded to conventional antiemetic treatments. Clinical formulations of dronabinol are available in capsule form (Marinol®), and as of July 2016 (with US FDA apporval), as an oral solution (Syndros®). Tetrahydrocannabinol, in a drug mixture containing cannabidiol (Sativex®, administered as an oromucosal spray), may also be used (in countries including the UK, Spain, Italy and Germany) to improve moderate to severe spastic symptoms in patients with multiple sclerosis (MS), and who have responded poorly to other anti-spasticity medications. The ability of dronabinol to reduce agitation in patients with Alzheimer's disease is being evaluated in clinical studies. |
Mechanism Of Action and Pharmacodynamic Effects  |
| The mechanism of action of THC is not completely understood. CB1 receptor agonist activity inhibits cyclic AMP production in areas of the central nervous system that mediate pain, memory, and other key functions, and is at least partially responsible for the physiological effects of cannabinoids. In addition, cannabidiol has been shown to stimulate vanilloid pain receptors (TRPVs), and to modulate cellular uptake and enzymatic hydrolysis of endogenous anandamide, effects which may contribute towards the pharmacological effects of cannabidiol [3]. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT02792257 | Trial of Dronabinol Adjunctive Treatment of Agitation in Alzheimer's Disease | Phase 2 Interventional | Johns Hopkins University | 4 | |
| NCT05239390 | SCI-110 for Alzheimer Disease and Agitation | Phase 2 Interventional | The Israeli Medical Center for Alzheimer's | ||
| NCT05612711 | Dronabinol for Agitation in Dementia Crossover Trial | Phase 2 Interventional | Ralph H. Johnson VA Medical Center | ||
| NCT05543681 | Clinical Trial on Agitation in Alzheimer's Dementia | Phase 2 Interventional | IGC Pharma, LLC | ||
External links  |
|
For extended ADME data see the following: Electronic Medicines Compendium (eMC) Drugs.com |