GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
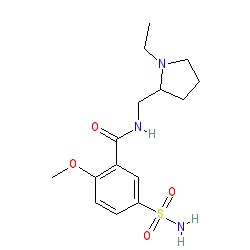
Synonyms: (±)-sulpiride | dl-sulpiride | Dolmatil® | RD 1403 | Sulpor® | sulpyrid
sulpiride is an approved drug
Compound class:
Synthetic organic
Comment: The approved drug sulpiride is an enantiomeric mixture of (+)-sulpiride and (-)-sulpiride. We show the non-isomeric structure to represent the mixture.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖
View more information in the IUPHAR Pharmacology Education Project: sulpiride |
|
|||||||||||||||||||||||||||||||||||
| References |
|
1. Auerbach SS, DrugMatrix® and ToxFX® Coordinator National Toxicology Program.
National Toxicology Program: Dept of Health and Human Services. Accessed on 02/05/2014. Modified on 02/05/2014. DrugMatrix, https://doi.org/10.22427/NTP-DATA-107-022-001-000-3 |
|
2. Burris KD, Pacheco MA, Filtz TM, Kung MP, Kung HF, Molinoff PB. (1995)
Lack of discrimination by agonists for D2 and D3 dopamine receptors. Neuropsychopharmacology, 12 (4): 335-45. [PMID:7576010] |
|
3. Sethi KK, Vullo D, Verma SM, Tanç M, Carta F, Supuran CT. (2013)
Carbonic anhydrase inhibitors: synthesis and inhibition of the human carbonic anhydrase isoforms I, II, VII, IX and XII with benzene sulfonamides incorporating 4,5,6,7-tetrabromophthalimide moiety. Bioorg Med Chem, 21 (19): 5973-82. [PMID:23965175] |
|
4. Vullo D, Innocenti A, Nishimori I, Pastorek J, Scozzafava A, Pastoreková S, Supuran CT. (2005)
Carbonic anhydrase inhibitors. Inhibition of the transmembrane isozyme XII with sulfonamides-a new target for the design of antitumor and antiglaucoma drugs?. Bioorg Med Chem Lett, 15 (4): 963-9. [PMID:15686894] |