GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Compound class:
Synthetic organic
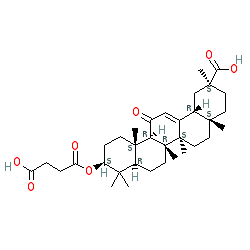
Comment: Carbenoxolone is derived from licorice root, and has a steroid-like chemical structure. Formulations used clinically typically contain carbenoxolone sodium.
|
|
|||||||||||||||||||||||||||||||||||
Classification  |
|
| Compound class | Synthetic organic |
IUPAC Name  |
| (2S,4aS,6aR,6aS,6bR,8aR,10S,12aS,14bR)-10-(4-hydroxy-4-oxobutanoyl)oxy-2,4a,6a,6b,9,9,12a-heptamethyl-13-oxo-3,4,5,6,6a,7,8,8a,10,11,12,14b-dodecahydro-1H-picene-2-carboxylic acid |
International Nonproprietary Names  |
|
| INN number | INN |
| 1789 | carbenoxolone |
Database Links  |
|
| CAS Registry No. | 5697-56-3 (source: Scifinder) |
| ChEMBL Ligand | CHEMBL499915 |
| DrugCentral Ligand | 493 |
| GtoPdb PubChem SID | 178100972 |
| PubChem CID | 636403 |
| RCSB PDB Ligand | CBO |
| Search Google for chemical match using the InChIKey | OBZHEBDUNPOCJG-WBXJDKIVSA-N |
| Search Google for chemicals with the same backbone | OBZHEBDUNPOCJG |
| Search PubMed clinical trials | carbenoxolone |
| Search PubMed titles | carbenoxolone |
| Search PubMed titles/abstracts | carbenoxolone |
| UniChem Compound Search for chemical match using the InChIKey | OBZHEBDUNPOCJG-WBXJDKIVSA-N |
| UniChem Connectivity Search for chemical match using the InChIKey | OBZHEBDUNPOCJG-WBXJDKIVSA-N |
| Wikipedia | Carbenoxolone |
Product suppliers
View disclaimer
Sponsored products disclaimer
Product supplier links are provided as a service to assist in identifying commercial suppliers of reagents that are mentioned on the IUPHAR/BPS Guide to PHARMACOLOGY database website, and do not imply their endorsement by NC-IUPHAR.
Links are provided in return for sponsorship, used to fund improvements to this database. The sponsorship account is managed and audited by the University of Edinburgh, a charitable body registered in Scotland, SC005336. If you are interested in sponsoring the database, please contact us.
✖
Sponsored products disclaimer
Product supplier links are provided as a service to assist in identifying commercial suppliers of reagents that are mentioned on the IUPHAR/BPS Guide to PHARMACOLOGY database website, and do not imply their endorsement by NC-IUPHAR.
Links are provided in return for sponsorship, used to fund improvements to this database. The sponsorship account is managed and audited by the University of Edinburgh, a charitable body registered in Scotland, SC005336. If you are interested in sponsoring the database, please contact us.
✖|
Carbenoxolone (links to external site)
Cat. No. HY-B1588 |