GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
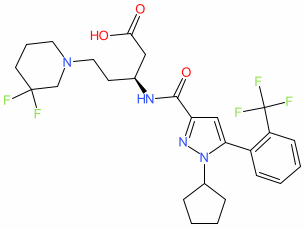
Synonyms: APT101 | compound 423 [WO2017100558A1] | compound 52 [PMID: 35594649]
Compound class:
Synthetic organic
Comment: APT-101 (APIE Therapeutics) is an orally bioavailable small molecule that acts as an apelin receptor (APLNR; APJ) agonist [2]. It activates the apelin/apelin receptor pathway to trigger the repair of pulmonary vascular endothelial cell breakdown that drives damage in fibrotic and sclerotic lung diseases [3,5-8].
We obtained the IUPAC descriptor for the molecule from the FDA Orphan Drug register. This chemical structure was first claimed in a patent series from RTI International [4], and is a structure match for compound 52 in [2]. |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| APT-101 was granted FDA orphan designation in 2022, to treat idiopathic pulmonary fibrosis (IPF) and systemic sclerosis [1]. |