GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
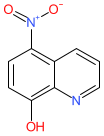
Synonyms: APL-1202 | APL1202 | compound 10 [PMID: 30939017]
nitroxoline is an approved drug
Compound class:
Synthetic organic
Comment: Nitroxoline is an antimicrobial used to treat urinary tract infections [3]. Repurposing of nitroxoline for infectious diseases and oncological indications has been proposed, although pharmacokinetic challenges are likely to hamper clinical translation.
From a patent it would appear that Healx Ltd. are interested in pursuing nitroxoline for the treatment of plexiform neurofibroma/neurofibromatosis type 1 (NF1) [1]. Review of Healx's active pipeline (January 2026) reveals two candidates with codes HLX-1502 and HLX-0213 in development for NF1 [4]. HLX-1502 has reached phase 2 whereas HLX-0213 is in the discovery phase (at time of writing), so it is plausible that HLX-1502 is nitroxoline. We await formal disclosure of this potential association. Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT06541847 | A Phase 2, Open-Label Study to Evaluate the Safety and Effects of HLX-1502 in Patients With Neurofibromatosis Type 1 | Phase 2 Interventional | Healx Limited | ||