GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: Analogue 6 [PMID: 37406381] | YK-029A
Compound class:
Synthetic organic
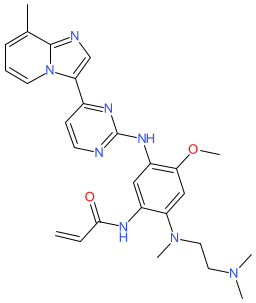
Comment: The chemical structure for pruvonertinib was derived from WHO proposed INN list 132 (Feb 2025), in which it is listed as an epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor with antineoplastic potential. A CAS number and structure match in PubChem linked the INN to the company research code YK-029A (Puhe Pharmaceutical). YK-029A was declared as an orally bioavailable EGFR inhibitor that targets receptors harbouring the T790M and exon 20 insertion (resistance) mutations in 2023 [1]. Structurally, pruvonertinib is an analogue derived from the osimertinib scaffold.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Pruvonertinib (YK-029A) is a clinical candidate for the treament of advanced NSCLC with confirmed sensitive EGFR alterations. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT05767892 | YK-029A as First-Line Treatment Versus Platinum-Based Chemotherapy for Non-Small Cell Lung Cancer (NSCLC) With EGFR Exon 20 Insertion Mutations | Phase 3 Interventional | Suzhou Puhe Pharmaceutical Technology Co., LTD | ||
| NCT05767866 | Assessing an Oral EGFR Inhibitor,YK-209A in Patients Who Have Advanced Non-small Cell Lung Cancer With EGFR | Phase 1/Phase 2 Interventional | Suzhou Puhe Pharmaceutical Technology Co., LTD | ||