GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Compound class:
Synthetic organic
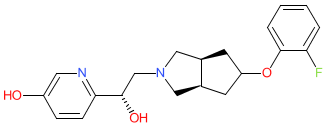
Comment: Onfasprodil was identified from WHO proposed INN list 126 (Jan 2022) as a N-methyl-D-aspartate (NMDA) receptor antagonist. It acts as a negative allosteric modulator of the glutamate ionotropic receptor NMDA type subunit 2B (GRIN2B; GluN2B or NR2B) and is proposed as an intravenous, rapid onset antidepressant. Onfasprodil's chemical structure is claimed in patent US10239835B2 [1]. We predict that is onfasprodil Novartis' Phase 2 clinical lead MIJ821, although name-to-structure has not been disclosed.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| Selectivity at ion channels | ||||||||||||||||||||||||||||||||||
| Key to terms and symbols | Click column headers to sort | |||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||