GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
Synonyms: compound 23a [PMID: 38670537] | TQB-3616 | TQB3616
culmerciclib is an approved drug
Compound class:
Synthetic organic
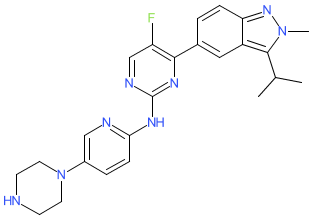
Comment: We obtained the chemical structure for culmerciclib from the WHO's proposed INN list 127 (21 July 2022). In this document it was described as a cyclin-dependent kinase inhibitor and antineoplastic. This Chemical structure mapped to PubChem CID 122544510, and to a structure in Nanjing Mingde New Drug Research and Development Company's patent WO2016141881A1 [1] which claims selective CDK4/6 inhibitors as anticancer drugs. A discolsure in late 2025, reported culmerciclib as a CDK2/4/6 inhibitor, and provided the synonym TQB3616 [5]. Discovery and preclinical evaluations TQB3616 had been reported in 2024 [4].
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Culmerciclib was approved by the Chinese NMPA in December 2025, to treat hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative locally advanced or metastatic breast cancer that has progressed following prior endocrine therapy, in combination with fulvestrant [3]. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT06702618 | Evaluation of TQB3616 Capsules in a Phase II Clinical Trial for Recurrent/Metastatic Breast Cancer | Phase 2 Interventional | Chia Tai Tianqing Pharmaceutical Group Co., Ltd. | ||
| NCT05375461 | TQB3616 Capsules Plus Fulvestrant Compared to Placebo Plus Fulvestrant in Previously Treated Breast Cancer in Clinical Trail | Phase 3 Interventional | Chia Tai Tianqing Pharmaceutical Group Co., Ltd. | 5 | |
| NCT05780567 | Clinical Study on Adjuvant Therapy of TQB3616 Combined With Endocrine Therapy Compared With Placebo Combined With Endocrine Therapy in Patients With Breast Cancer | Phase 3 Interventional | Chia Tai Tianqing Pharmaceutical Group Co., Ltd. | ||
| NCT05365178 | To Evaluate the Efficacy and Safety of TQB3616 in Combination With Fulvestrant Versus Placebo in Combination With Fulvestrant in Previously Untreated Hormone-receptor (HR)-Positive, Human Epidermal Growth Factor Receptor 2 (HER2)-Negative Advanced Breast Cancer | Phase 3 Interventional | Chia Tai Tianqing Pharmaceutical Group Co., Ltd. | ||