GtoPdb is requesting financial support from commercial users. Please see our sustainability page for more information.
|
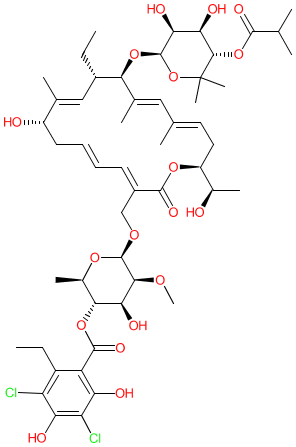
Synonyms: Dificid® | Dificlir® | lipiarmycin | OPT-80 | PAR-101 | tiacumicin B
fidaxomicin is an approved drug (EMA & FDA (2011))
Compound class:
Synthetic organic
Comment: Fidaxomicin is a narrow-spectrum, semisynthetic, macrolide antibacterial. It was isolated from the hamdenensis subspecies of actinomycete Dactylosporangium aurantiacum.
|
|
|||||||||||||||||||||||||||||||||||
| No information available. |
External links  |
|
For extended ADME data see the following: Electronic Medicines Compendium (eMC) Drugs.com European Medicines Agency (EMA) |