|
Compound class:
Synthetic organic
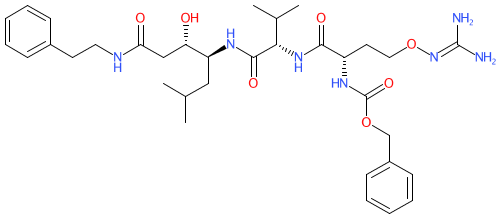
Comment: WEHI-842 was developed as a potent peptidomimetic inhibitor of Plasmodium plasmepsin V (PMV), a promising target for antimalarial drug development [2].
The Malaria tab on this ligand page provides additional curator comments of relevance to the Guide to MALARIA PHARMACOLOGY. Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| Guide to Malaria Pharmacology Comments |
| Future progression of peptidomimetic Plasmodium plasmepsin V inhibitors, such as WEHI-842, through the antimalarial drug development pipeline will require improvements to their biochemical and pharmacokinetic properties. The 100-fold reduction in potency observed between isolated enzyme and cultured parasites could lead to unacceptable dosing levels in vivo. This loss of potency in cell-based assays may be explained by the finding, from functional genetics studies, that nearly all cellular PMV must be inhibited before parasite-killing effects are observed [4]. Patent WO2016197190 relates to a class of compounds based on WEHI-842 [1]. Potential Target/Mechanism Of Action: WEHI-842 inhibits Plasmodium PMV (see Clinical data tab for further details). |