|
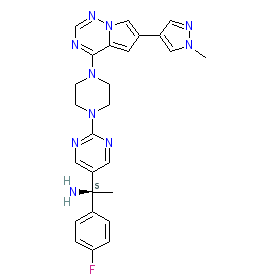
Synonyms: Ayvakit® | Ayvakyt® | BLU-285 | BLU285
avapritinib is an approved drug (FDA & EMA (2020))
Compound class:
Synthetic organic
Comment: Avapritinib (BLU-285) was developed by Blueprint Medicines as an oral precision therapy to selectively and potently inhibit KIT and PDGFRA mutant kinases [3]. It is a Type I kinase inhibitor that binds the active kinase conformation. Avapritinib binds to the 'switch pocket' of KIT, a domain that regulates the enzyme's catalytic conformation. Compounds that act in this way are often referred to as switch control inhibitors [1]. Another such switch control inhibitor is ripretinib.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| Immunopharmacology Comments |
| A constitutively active mutant KIT (typically D816V) is present in 90-95% of cases of systemic mastocytosis and drives disease pathogenesis. The multi-kinase inhibitor midostaurin is currently the only approved treatment for advanced/aggressive systemic mastocytosis [4]. Avapritinib was designed to directly target the active conformation of KIT, so as to potently and selectively inhibit KITD816V and other activation loop mutants [3] that confer resistance to early generation kinase inhibitors such as imatinib. |