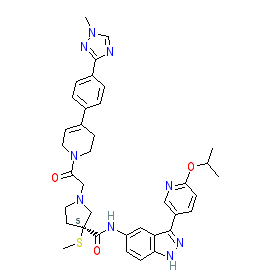
|
Synonyms: MK8353 | SCH900353
Compound class:
Synthetic organic
Comment: MK-8353 (formerly know as SCH900353) is an orally bioavailable dual mechanism ERK1/2 inhibitor, i.e. it inhibits ERK1/2 catalytic activity and phosphorylation of ERK1/2 [1]. It has improved pharmacokinetic properties in comparison to SCH772984 (a preclinical ERK1/2 inhibtor tool compound) and has shown preliminary anti-tumour efficacy in patients with BRAF or RAS mutated metastatic melanoma and colorectal cancers.
(Note: ERK1= MAPK3, ERK2= MAPK1). Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| A Phase 1 study of MK-8353 as a single agent in patients with advanced solid tumours (NCT01358331) was terminated; results from this trial were published by MOschos et al. (2018) [1]. ClinicalTrials.gov has trial number NCT01358331 listed as 'recruiting', and this study is designed to evaluate the eficacy of MK-8353 in combination with the approved PD-1 checkpoint inhibitor pembrolizumab, in patients with advanced colorectal cancer. |
| Clinical Trials | |||||
| Clinical Trial ID | Title | Type | Source | Comment | References |
| NCT01358331 | A Study of the Safety, Tolerability, and Efficacy of MK-8353 in Participants With Advanced Solid Tumors (MK-8353-001) | Phase 1 Interventional | Merck Sharp & Dohme Corp. | ||
| NCT02972034 | Study of MK-8353 in Combination With Pembrolizumab (MK-3475) in Participants With Advanced Malignancies (MK-8353-013) | Phase 1 Interventional | Merck Sharp & Dohme Corp. | ||