|
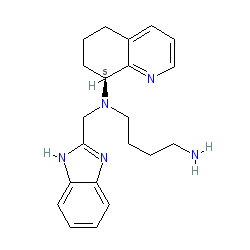
Synonyms: AMD 070 | AMD-070 | AMD-11070 | AMD11070 | compound 2 [PMID: 20297846] | X4P-001 | X4P-001-IO | X4P-001-LD
Compound class:
Synthetic organic
Comment: Mavorixafor (AMD070) is a potent, selective and bioavailable CXCR4 chemokine receptor allosteric antagonist [1]. Originally developed for HIV treatment, it is now being repurposed by X4 Pharmaceuticals as X4P-001 for the treatment of WHIM syndrome, a sub-type of a primary immunodeficiency disease caused by CXCR4 mutations. The compound is exemplified in a process patent US7332605 and as compound 89 from a series of 169 analogues in WO2003055876 but neither filing includes activity data.
Ligand Activity Visualisation ChartsThese are box plot that provide a unique visualisation, summarising all the activity data for a ligand taken from ChEMBL and GtoPdb across multiple targets and species. Click on a plot to see the median, interquartile range, low and high data points. A value of zero indicates that no data are available. A separate chart is created for each target, and where possible the algorithm tries to merge ChEMBL and GtoPdb targets by matching them on name and UniProt accession, for each available species. However, please note that inconsistency in naming of targets may lead to data for the same target being reported across multiple charts. ✖ |
|
|||||||||||||||||||||||||||||||||||
| No information available. |
Summary of Clinical Use  |
| Mavorixafor is being evaluated in clinical trials in patients with WHIM syndrome and in neutropenia. Click here to link to ClinicalTrials.gov's full list of mavorixafor studies. A Phase 3 trial was recruiting WHIM syndrome patients as of November 2019. This drug has Orphan Designation in the US (October 2018) and EU (July 2019) as a treament for WHIM syndrome. In November 2019 the US FDA granted mavorixafor Breakthrough Therapy Designation for this rare indication. |